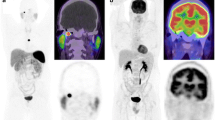
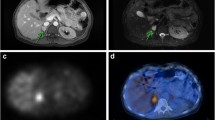
Abstract
Neuroectodermal tumours arise from chromaffin cells and possess the ability to secrete catecholamines. They are generally rare and may occur in association with a variety of hereditary syndromes such as MEN-2A and 2B, neurofibromatosis type 1 and von Hippel-Lindau disease. The most common types are phaeochromocytoma arising from the adrenal medulla and paraganglioma of extra-adrenal origin. Phaeochromocytomas tend to be benign and are often associated with a gene mutation if the disease is bilateral, while paragangliomas are often malignant, have a more aggressive nature and tend to metastasize. There are no specific histological or immunohistochemical features that indicate the malignant potential and the diagnosis of malignancy can only be established by the presence of distant metastases. Therefore, imaging can play a vital role in the diagnosis, localization, staging and assessment of spread. Traditionally, this is achieved with a combination of cross-sectional (CT and MRI) and functional (123I-MIBG or 111In-octreotide) imaging. However, these modalities are not adequate and achieve moderate sensitivity. The introduction of 68Ga-DOTA peptide in PET/CT imaging has led to improved receptor targeting and superb PET resolution, as well as accurate localization of lesions. The use of this technique in neuroectodermal tumours has been shown to be superior to all available modalities, but the available data are limited and larger studies are awaited to establish its role in the management of these tumours.

Similar content being viewed by others
References
Baysal BE. Hereditary paraganglioma targets diverse paraganglia. J Med Genet. 2002;39:617–22.
Neumann HP, Bausch B, McWhinney SR, Bender BU, Gimm O, Franke G, et al. Germline mutations in nonsyndromic pheochromocytoma. N Eng J Med. 2002;346:1459–66.
Lenders JW, Eisenhofer G, Mannelli M, Pacak K. Phaeochromocytoma. Lancet. 2005;366:665–75.
Baysal BE, Ferell RE, Willet-Brozick JE, Lawrence EC, Myssiorek D, Bosch A, et al. Mutations in SDHD, a mitochondrial complex II gene, in hereditary paragangliomas. Science. 2000;287:848–51.
Boedeker CC, Ridder GJ, Schipper J. Paragangliomas of the head and neck: diagnosis and treatment. Fam Cancer. 2005;4:55–9.
Persky MS, Setton A, Niimi Y, Hartman J, Frank D, Berenstein A. Combined endovascular and surgical treatment of head and neck paragangliomas: a team approach. Head Neck. 2002;24:423–31.
Ilias I, Pacak K. Current approaches and recommended algorithm for the diagnostic localization of pheochromocytoma. J Clin Endocrinol Metab. 2004;89:479–91.
Maurea S, Cuocolo A, Reynolds JC, Tumeh SS, Begley MG, Linehan WM, et al. Iodine-131-metaiodobenzylguanidine scintigraphy in preoperative and postoperative evaluation of paragangliomas: comparison with CT and MRI. J Nucl Med. 1993;34:173–9.
Go AS. Refining probability: an introduction to the use of diagnostic tests. In: Friedland DJ, editor. Evidence-based medicine. New York: McGraw-Hill; 1998. p. 12–33.
Schulz C, Eisenhofer G, Lehnert H. Principles of catecholamine biosynthesis, metabolism and release. Front Horm Res. 2004;31:1–25.
Bomanji J, Levison DA, Flatman WD, Horne T, Bouloux PM, Ross G, et al. Uptake of iodine-123 MIBG by pheochromocytomas, paragangliomas, and neuroblastomas: a histopathological comparison. J Nucl Med. 1987;28:973–8.
Furuta N, Kiyota H, Yoshigoe F, Hasegawa N, Ohishi Y. Diagnosis of pheochromocytoma using [123I]- compared with [131I]-metaiodobenzylguanidine scintigraphy. Int J Urol. 1999;6:119–24.
Shulkin BL, Ilias I, Sisson JC, Pacak K. Current trends in functional imaging of pheochromocytomas and paragangliomas. Ann N Y Acad Sci. 2006;1073:374–82.
Berglund AS, Hulthen UL, Manhem P, Thorsson O, Wollmer P, Törnquist C, et al. Metaiodobenzylguanidine (MIBG) scintigraphy and computed tomography (CT) in clinical practice. Primary and secondary evaluation for localization of phaeochromocytomas. J Intern Med. 2001;249:247–51.
Mann GN, Link JM, Pham P, Pickett CA, Byrd DR, Kinahan PE, et al. [11C]metahydroxyephedrine and [18F]fluorodeoxyglucose positron emission tomography improve clinical decision making in suspected pheochromocytoma. Ann Surg Oncol. 2006;13(2):187–97.
Kwekkeboom D, van Urk H, Pauw KH, Lamberts SW, Kooij PP, Hoogma RP, et al. Octreotide scintigraphy for the detection of paraganglioma. J Nucl Med. 1993;34:873–8.
van der Harst E, de Herder WW, Bruining HA, Bonjer HJ, de Krijger RR, Lamberts SW, et al. [(123)I]metaiodobenzylguanidine and [(111)In]octreotide uptake in benign and malignant pheochromocytomas. J Clin Endocrinol Metab. 2001;86:685–93.
Koopmans KP, Jager PL, Kema IP, Kerstens MN, Albersy F, Dullaart RP. 111In-octreotide is superior to 123I-metaiodobenzylguanidine for scintigraphic detection of head and neck paragangliomas. J Nucl Med. 2008;49:1232–7.
Bombardieri E, Seregni E, Villano C, Chiti A, Bajetta E. Position of nuclear medicine techniques in the diagnostic work-up of neuroendocrine tumors. Q J Nucl Med Mol Imaging. 2004;48:150–63.
Ueberberg B, Tourne H, Redman A, Walz MK, Schmid KW, Mann K, et al. Differential expression of the human somatostatin receptor subtypes sst1 to sst5 in various adrenal tumours and normal adrenal gland. Horm Metab Res. 2005;37:722–8.
Neumann DR, Basile KE, Bravo EL, Chen EQ, Go RT. Malignant pheochromocytoma of the anterior mediastinum: PET findings with [18F]FDG and 82Rb. J Comput Assist Tomogr. 1996;20:312–6.
Ilias I, Sahdev A, Reznek RH, Grossman AB, Pacak K. The optimal imaging of adrenal tumours: a comparison of different methods. Endocr Relat Cancer. 2007;14:587–99.
Mottaghy FM, Reske SN. Functional imaging of neuroendocrine tumours with PET. Pituitary. 2006;9:237–42.
Brink I, Hoegerle S, Klisch J, Bley TA. Imaging of pheochromocytoma and paraganglioma. Fam Cancer. 2005;4(1):61–8.
Rosenspire KC, Haka MS, Van Dort ME, Jewett DM, Gildersleeve DL, Schwaiger M, et al. Synthesis and preliminary evaluation of 11C metahydroxyephedrine: a false neurotransmitter agent for heart neuronal imaging. J Nucl Med. 1990;31:1328–34.
Shulkin BL, Wieland DM, Schwaiger M, Thompson NW, Francis IR, Haka MS, et al. PET scanning with hydroxyephedrine: an approach to the localization of phaeochromocytoma. J Nucl Med. 1992;33(6):1125–31.
Trampal C, Engler H, Juhlin C, Bergström M, Långström B. Pheochromocytomas: detection with 11C hydroxyephedrine PET. Radiology. 2004;230(2):423–8.
Ilias I, Yu J, Carrasquillo JA, Chen CC, Eisenhofer G, Whatley M, et al. Superiority of 6-[18F]-fluorodopamine positron emission tomography versus [131I]-metaiodobenzylguanidine scintigraphy in the localization of metastatic pheochromocytoma. J Clin Endocrinol Metab. 2003;88:4083–7.
Ilias I, Chen CC, Carrasquillo JA, Whatley M, Ling A, Lazúrová I, et al. Comparison of 6-18F-fluorodopaime PET with I-123 metaiodobenzylguanidine and In-111 pentetreotide scintigraphy in localisation of non metastatic and metastatic phaeochromocytoma. J Nucl Med. 2008;49:1613–9.
Kaji P, Carrasquillo JA, Linehan WM, Chen CC, Eisenhofer G, Pinto PA, et al. The role of 6-[18F]fluorodopamine positron emission tomography in the localization of adrenal pheochromocytoma associated with von Hippel-Lindau syndrome. Eur J Endocrinol. 2007;156:483–7.
Morrish PK, Rakshi JS, Bailey DL, Sawle GV, Brooks DJ. Measuring the rate of progression and estimating the preclinical period of Parkinson’s disease with [18F] dopa PET. J Neurol Neurosurg Psychiatry. 1998;64:314–9.
Hoegerle S, Nitzsche E, Altehoefer C, Ghanem N, Manz T, Brink I, et al. Pheochromocytomas: detection with 18F DOPA whole body PET – initial results. Radiology. 2002;222:507–12.
Timmers H, Chen C, Carrasquillo J, Whatley M, Ling A, Havekes B, et al. Comparison of 18F-fluoro-L-DOPA, 18F-fluoro-deoxyglucose, and 18F-fluorodopamine PET and 123I-MIBG scintigraphy in the localization of pheochromocytoma and paraganglioma. J Clin Endocrinol Metab. 2009;94(12):4757–67.
Reynolds S, Lewington V. Radionuclide imaging of phaeochromocytoma and paraganglioma. Imaging. 2008;3(4):21–4.
Rufini V, Calcagni ML, Baum RP. Imaging of neuroendocrine tumours. Semin Nucl Med. 2006;36:228–47.
Ambrosini V, Rubello D, Nanni C, Al Nahhas A, Fanti S. 68Ga-DOTA-peptides versus 18F-DOPA PET for the assessment of NET patients. Nucl Med Commun. 2008;29:415–7.
Win Z, Al-Nahhas A, Towey D, Todd JF, Rubello D, Lewington V, et al. 68Ga-DOTATATE PET in neuroectodermal tumours: first experience. Nucl Med Commun. 2007;28:359–63.
Arnold R, Tautmann ME, Creutzfeldt W, Benning R, Benning M, Neuhaus C, et al. Somatostatin analogue octreotide and inhibition of tumour growth in metastatic endocrine gastroenteropancreatic tumours. Gut. 1996;38:430–8.
Wild D, Mäcke HR, Waser B, Reubi JC, Ginj M, Rasch H. 68Ga-DOTANOC: a first compound for PET imaging with high affinity for somatostatin receptor subtypes 2 and 5. Eur J Nucl Med Mol Imaging. 2005;32:724.
Wild D, Schmitt JS, Ginj M, Mäcke HR, Bernard BF, Krenning E. DOTA-NOC, a high-affinity ligand of somatostatin receptor subtypes 2, 3 and 5 for labelling with various radiometals. Eur J Nucl Med Mol Imaging. 2003;30:1338–47.
Ginj M, Chen J, Walter MA, Eltschinger V, Reubi JC, Maecke HR. Preclinical evaluation of new and highly potent analogues of octreotide for predictive imaging and targeted radiotherapy. Clin Cancer Res. 2005;11:1136–45.
Fanti S, Ambrosini V, Tomassetti P, Castellucci P, Montini G, Allegri V, et al. Evaluation of unusual neuroendocrine tumours by means of 68Ga-DOTA-NOC PET. Biomed Pharmacother. 2008;62:667–71.
Miederer M, Molatore S, Marinoni I, Perren A, Spitzweg C, Reder S, et al. Functional imaging of pheochromocytoma with Ga-DOTATOC and C-HED in a genetically defined rat model of multiple endocrine neoplasia. Int J Mol Imaging. 2011;2011:17532. doi:10.1155/2011/175352.
Kroiss A, Putzer D, Uprimny C, Decristoforo C, Gabriel M, Santner W, et al. Functional imaging in phaeochromocytoma and neuroblastoma with 68Ga-DOTA-Tyr 3-octreotide positron emission tomography and 123I-metaiodobenzylguanidine. Eur J Nucl Med Mol Imaging. 2011;38(5):865–73.
Kwekkeboom DJ, de Herder WW, Kam BL, van Eijck CH, van Essen M, Kooij PP, et al. Treatment with the radiolabeled somatostatin analog [177Lu-DOTA 0,Tyr3]octreotate: toxicity, efficacy, and survival. J Clin Oncol. 2008;26:2124–30.
Frilling A, Sotiropoulos GC, Radtke A, Malago M, Bockisch A, Kuehl H, et al. The impact of 68Ga-DOTATOC positron emission tomography/computed tomography on the multimodal management of patients with neuroendocrine tumors. Ann Surg. 2010;252(5):850–6.
Win Z, Rahman L, Murrell J, Todd J, Al-Nahhas A. The possible role of 68Ga-DOTATATE PET in malignant abdominal paraganglioma. Eur J Nucl Med Mol Imaging. 2006;33(4):506.
Naji M, Zhao C, Welsh SJ, Meades R, Win Z, Ferrarese A, et al. 68Ga-DOTA-TATE PET vs. 123I-MIBG in identifying malignant neural crest tumours. Mol Imaging Biol. 2011;13(4):769–75.
Farnsworth J, Alsayed M, Zerizer I, Naji M, Meades R, Williams B. Retrospective study comparing Ga-68 DOTATATE PET with co-registered low dose CT in the detection of bone metastases in patients with neuroendocrine tumours. Nucl Med Commun. 2011;32:426. Abstract.
Putzer D, Gabriel M, Henninger B, Kendler D, Uprimny C, Dobrozemsky G, et al. Bone metastases in patients with neuroendocrine tumor: 68Ga-DOTA-Tyr3-octreotide PET in comparison to CT and bone scintigraphy. J Nucl Med. 2009;50(8):1214–21.
Conflicts of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Naji, M., AL-Nahhas, A. 68Ga-labelled peptides in the management of neuroectodermal tumours. Eur J Nucl Med Mol Imaging 39 (Suppl 1), 61–67 (2012). https://doi.org/10.1007/s00259-011-1990-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00259-011-1990-y