Abstract
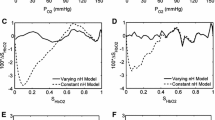
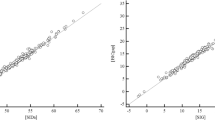
Mathematical models of the acid–base chemistry of blood based upon mass action and mass balance equations have become popular as diagnostic tools in intensive care. The reference models using this approach are those based on the strong ion approach, but these models do not currently take into account the effects of oxygen on the buffering characteristics of haemoglobin. As such these models are limited in their ability to simulate physiological situations involving simultaneous changes of O2 and CO2 levels in the blood. This paper describes a model of acid–base chemistry of blood based on mass action and mass balance equations and including the effects of oxygen. The model is used to simulate the mixing of venous blood with the same blood at elevated O2 and reduced CO2 levels, and the results compared with the mixing of blood sampled from 21 healthy subjects. Simulated values of pH, PCO2, PO2 and SO2 in the mixed blood compare well with measured values with small bias (i.e. 0.000 pH, −0.06 kPa PCO2, −0.1% SO2, −0.02 kPa PO2), and values of standard deviations (i.e. 0.006 pH, 0.11 kPa PCO2, 0.8% SO2, 0.13 kPa PO2) comparable to the precision seen in direct measurement of these variables in clinical practice. These results indicate that the model can reliably simulate the mixing of blood and has potential for application in describing physiological situations involving the mixing of blood at different O2 and CO2 levels such as occurs in the mixing of lung capillary and shunted pulmonary blood.



Similar content being viewed by others
References
Allerød C, Rees SE, Rasmussen BS, Karbing DS, Kjærgaard S, Thorgaard P, Andreassen SA (2008) Decision support system for suggesting ventilator settings: Retrospective evaluation in cardiac surgery patients ventilated in the ICU. Comput Methods Programs Biomed 92:205–212
Constable PD (1997) A simplified strong ion model for acid–base equilibria: application to horse plasma. J Appl Physiol 83:297–311
Constable PD (2001) Total weak acid concentration and effective dissociation constrant of nonvolatile buffers in human plasma. J Appl Physiol 91:1364–1371
Constable PD (2003) Hyperchloremic acidosis: the classic example of strong ion acidosis. Anesth Analg 96:919–922
Dubin A, Menises MM, Masevicius FD, Moseinco MC, Kutscherauer DO, Ventrice E, Laffaire E, Estenssoro E (2007) Comparison of three different methods of evaluation of metabolic acid–base disorders. Crit Care Med 35:1264–1270
Duffin J (2005) The role of acid–base balance in the chemoreflex control of breathing. J Appl Physiol 99:2255–2265
Figge J, Jabor A, Kazda A, Fencl V (1998) Anion gap and hypoalbuminemia. Crit Care Med 26:1807–1810
Funder J, Weith JO (1966) Chloride and hydrogen ion distribution between human red cells and plasma. Acta Physiol Scand 68:234–245
Honore PM, Joannes-Boyau O, Boer W (2009) Strong ion gap and outcome after cardiac arrest: another nail in the coffin of traditional acid–base quantification. Intensive Care Med 35:189–191
Lindinger MI, Kowalchuk JM, Heigenhauser GJF (2005) Applying physiochemical principles to skeletal muscle acid–base status. Am J Physiol Regul Integr Comp Physiol 289:R891–R894
McKelvie RS, Lindinger MI, Jones NL, Heigenhauser GJF (1992) Erythrocyte ion regulation across inactive muscle during leg exercise. Can J Physiol Pharmacol 70:1625–1633
Morgan TJ (2005) The meaning of acid–base abnormalities in the intensive care unit-effects of fluid administration. Critical Care 9:204–211
Radiometer Medical A/S (2004) ABL 800 Flex Reference Manual
Rees SE, Andreassen S (2005) Mathematical models of oxygen and carbon dioxide storage and transport: the acid–base chemistry of blood. Crit Rev Biomed Eng 33:209–264
Rees SE, Allerød C, Murley D, Zhao Y, Smith BW, Kjærgaard S, Thorgaard P, Andreassen S (2006a) Using physiological models and decision theory for selecting appropriate ventilator settings. J Clin Monit Comput 20:421–429
Rees SE, Toftegaard M, Andreassen S (2006b) A method for calculation of arterial acid–base status from measurements in the peripheral venous blood. Comput Methods Programs Biomed 81:18–25
Rees SE, Hansen A, Toftegaard M, Pedersen J, Kristiensen SR, Harving H (2009) Converting venous acid–base and oxygen status to arterial in patients with lung disease. Eur Respir J 33:1141–1147
Schwartz WB, Relman AS (1963) A critique of the parameters used in the evaluation of acid–base disorders “whole-blood buffer base” and “standard bicarbonate” compared with blood pH and plasma bicarbonate concentration. N Engl J Med 268:1382–1388
Siggaard-Andersen O (1974) The acid–base status of the blood. Munksgaard, Copenhagen
Siggaard-Andersen M, Siggaard-Andersen O (1995) Oxygen status algorithm, version 3, with some applications. Acta Anaesthsiol Scand 39(S107):13–20
Siggaard-Andersen O, Wimberly PD, Gøthgen I, Siggaard-Andersen M (1984) A mathematical model of the haemoglobin-oxygen dissociation curve of the human blood and of the oxygen partial pressure as a function of temperature. Clin Chem 30:1646–1651
Siggaard-Andersen O, Wimberly PD, Fogh-Andersen N, Gøthgen I (1988) Measured and derived quantities with modern pH and blood gas equipment: calculation algorithms with 54 equations. Scand J Clin Lab Invest 48(S189):7–15
Singer RB, Hastings AB (1948) An improved clinical method for the estimation of disturbances of the acid–base balance of human blood. Medicine 27:223–242
Stewart PA (1983) Modern quantitative acid–base chemistry. Can J Physiol Pharmacol 61:1444–1461
Toftegaard M, Rees SE, Andreassen S (2009) Evaluation of a method for converting venous values of acid–base and oxygenation status to arterial values. Emerg Med J 26:268–272
Wooten EW (1999) Analytic calculation of physiological acid–base parameters in plasma. J Appl Physiol 86:326–334
Wooten EW (2003) Calculation of physiological acid–base parameters in multicompartment systems with application to human blood. J Appl Physiol 95:2333–2344
Author information
Authors and Affiliations
Corresponding author
Additional information
Communicated by Susan Ward.
Rights and permissions
About this article
Cite this article
Rees, S.E., Klæstrup, E., Handy, J. et al. Mathematical modelling of the acid–base chemistry and oxygenation of blood: a mass balance, mass action approach including plasma and red blood cells. Eur J Appl Physiol 108, 483–494 (2010). https://doi.org/10.1007/s00421-009-1244-x
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00421-009-1244-x