Abstract
Goals of work
The aims of the present study were to verify whether an innovative therapeutic strategy for the treatment of mild-moderate chronic cancer pain, passing directly from step I to step III of the WHO analgesic ladder, is more effective than the traditional three-step strategy and to evaluate the tolerability and therapeutic index in both strategies.
Methods
Patients aged 18 years or older with multiple viscera or bone metastases or with locally advanced disease were randomized. Pain intensity was assessed using a 0–10 numerical rating scale based on four questions selected from the validated Italian version of the Brief Pain Inventory. Treatment-specific variables and other symptoms were recorded at baseline up to a maximum follow-up of 90 days per patient.
Results
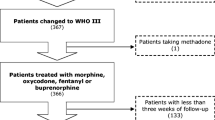
Fifty-four patients were randomized onto the study, and pain intensity was assessed over a period of 2,649 days. The innovative treatment presented a statistically significant advantage over the traditional strategy in terms of the percentage of days with worst pain ≥5 (22.8 vs 28.6%, p<0.001) and ≥7 (8.6 vs 11.2%, p=0.023). Grades 3 and 4 anorexia and constipation were more frequently reported in the innovative strategy arm, although prophylactic laxative therapy was used less in this setting.
Conclusions
Our preliminary data would seem to suggest that a direct move to the third step of the WHO analgesic ladder is feasible and could reduce some pain scores but also requires careful management of side effects.

Similar content being viewed by others
References
American Pain Society Quality of Care Committee (1995) Quality improvement guidelines for the treatment of acute pain and cancer pain. JAMA 274:1874–1880
Ashby MA, Fleming BG, Brooksbank M et al (1992) Description of a mechanistic approach to pain management in advanced cancer. Preliminary report. Pain 51:153–161
Bonica JJ (1990) Cancer pain. In: Bonica JJ (ed) The management of pain, 2nd edn. Lea & Febiger, Philadelphia, PA
Brooks DJ, Gamble W, Ahmedzai S (1995) A regional survey of opioid use by patients receiving specialist palliative care. Palliat Med 9:229–238
Cherny NI, Portenoy RK (1994) Practical issues in the management of cancer pain. In: Wall PD, Melzach R (eds) Textbook of pain. Churchill Livingstone, London, UK, pp 1437–1468
Cleeland CS, Gonin R, Hatfield AK et al (1994) Pain and its treatment in outpatients with metastatic cancer. N Engl J Med 330:592–596
Du Pen SL, Du Pen AR, Polissar N et al (1999) Implementing guidelines for cancer pain management: results of a randomized controlled clinical trial. J Clin Oncol 17:361–370
Eisemberg E, Berkey CS, Carr DB et al (1994) Efficacy and safety of nonsteroidal antiinflammatory drugs for cancer pain: a meta-analysis. J Clin Oncol 12:2756–2765
Freynhagen R, Zenz M, Strumpf M (1994) WHO step II: clinical reality or a didactic instrument? Schmerz 8:210–215
Grond S, Radbruch L, Menser T (1999) High-dose tramadol in comparison to low-dose morphine for cancer pain relief. J Pain Symptom Manage 18:174–179
ICH Harmonised Tripartite Guideline (1998) Statistical principles for clinical trials. The European Agency for the Evaluation of Medicinal Products, London, pp 22–24
Jadad AR, Browman GP (1995) The WHO analgesic ladder for cancer pain management. Stepping up the quality of its evaluation. JAMA 274:1870–1873
Kumar KS, Rajagopal MR, Naseema AM (2000) Intravenous morphine for emergency treatment of cancer pain. Palliat Med 14:183–188
Laudico AV (1995) Cancer pain relief: a primary health care issue in the Philippines. Cancer Pain Release 8:1–4
Marinangeli F, Ciccozzi A, Leonardis M et al (2004) Use of strong opioids in advanced cancer pain: a randomized trial. J Pain Symptom Manage 27:409–416
Mercadante S, Salvaggio L, Dardanoni G et al (1998) Dextropropoxyphene versus morphine in opioid-naive cancer patients with pain. J Pain Symptom Manage 15:76–81
Meuser T, Pietruck C, Radbruch L, Stute P, Lehmann KA, Grond S (2001) Symptoms during cancer pain treatment following WHO-guidelines: a longitudinal follow-up study of symptom prevalence, severity and etiology. Pain 93:247–257
Minotti V, de Angelis V, Rigetti E et al (1998) Double blind evaluation of short-term analgesic efficacy of orally administered diclofenac, diclofenac plus codeine, and diclofenac plus imipramina in chronic cancer pain. Pain 74:133–137
Porta-Sales J, Gòmez-Batiste X, Tuca-Rodriguez A, Madrid-Juan F, Espinosa-Rojas J, Trelis-Navarro J (2003) WHO analgesic ladder-or lift? Eur J Palliat Care 10:105–109
Regnard CFB, Tempest S (1992) A guide to symptom relief in advanced cancer. Haigh and Hochland, Manchester, UK
SAS/SAT (1990) User’s Guide, version 8.02. SAS Institute, Cary, NC
Serlin RC, Mendoza TR, Nakamura Y, Edwards KR, Cleeland CS (1995) When is cancer pain mild, moderate or severe? Grading pain severity by its interference with function. Pain 61:277–284
Stjernswärd J, Teoh N (1990) The scope of the cancer pain problem. In: Foley KM, Bonica JJ, Ventafridda V, Callaway MV (eds) Advances in pain research and therapy. Raven Press, New York, NY, pp 7–12
Ventafridda V, Tamburini M, Caraceni A, De Conno F, Naldi F (1987) A validation study of the WHO method for cancer pain relief. Cancer 59:850–856
Vielvoye-Kerkmcer APE, Mattern C, Vitendaal MP (2000) Transdermal fentanyl in opioid-naive cancer pain patients: an open trial using transdermal fentanyl for the treatment of chronic cancer pain in opioid-naive patients and a group using codeine. J Pain Symptom Manage 19:185–192
World Health Organization (1986) Cancer pain relief. World Health Organization, Geneva, Switzerland
World Health Organization (1990) Cancer pain relief and palliative care: report of a WHO expert committee. Technical report series 804. World Health Organization, Geneva, Switzerland
World Health Organization (1996) Cancer pain relief: with a guide to opioid availability. World Health Organization, Geneva, Switzerland
Zech DFJ, Grond S, Lynch J, Hertel D, Lehmann KA (1995) Validation of World Health Organization guidelines for cancer pain relief: a 10-year prospective study. Pain 63:65–76
Acknowledgements
The co-authorship of all the following group members is acknowledged: Laura Fabbri (Palliative Care Unit, Forlimpopoli Hospital, Forlimpopoli), Oriana Nanni (Unit of Biostatistics and Clinical Trials, Istituto Oncologico Romagnolo, Forlì), Paola Raulli (Palliative Care Unit, Bassini Hospital, Cinisello Balsamo), Barbara Poggi and Francesca Fochessati (Department of Oncology, Infermi Hospital, Rimini), Donatella Giannunzio and Maria Lucia Barbagallo (Palliative Care Unit, Azienda Istituti Ospitalieri, Cremona), Vincenzo Minotti and Maura Betti (Department of Oncology, Monteluce Polyclinic, Perugia), Stefano Giordani (Hospice-ADO Casa della Solidarietà, Ferrara), and Elena Piazza, Roberto Scapaticci, and Sabrina Ferrario (Department of Oncology, Luigi Sacco Hospital, Milan). We thank Gráinne Tierney for editing the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Maltoni, M., Scarpi, E., Modonesi, C. et al. A validation study of the WHO analgesic ladder: a two-step vs three-step strategy. Support Care Cancer 13, 888–894 (2005). https://doi.org/10.1007/s00520-005-0807-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00520-005-0807-6