Abstract
Hepatic encephalopathy (HE) is a serious neuropsychiatric complication of both acute and chronic liver disease. Symptoms of HE can include confusion, disorientation and poor coordination. A general consensus exists that the synergistic effects of excess ammonia and inflammation cause astrocyte swelling and cerebral edema; however, the precise molecular mechanisms that lead to these morphological changes in the brain are unclear. Cerebral edema occurs to some degree in all patients with HE, regardless of its grade, and could underlie the pathogenesis of this disorder. The different grades of HE can be diagnosed by a number of investigations, including neuropsychometric tests (such as the psychometric hepatic encephalopathy score), brain imaging and clinical scales (such as the West Haven criteria). HE is best managed by excluding other possible causes of encephalopathy alongside identifying and the precipitating cause, and confirming the diagnosis by a positive response to empiric treatment. Empiric therapy for HE is largely based on the principle of reducing the production and absorption of ammonia in the gut through administration of pharmacological agents such as rifaximin and lactulose, which are approved by the FDA for the treatment of HE.
Key Points
-
Hepatic encephalopathy (HE) is a serious neuropsychiatric complication of acute and chronic liver disease
-
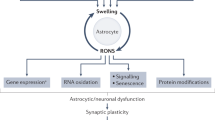
Inflammation and raised levels of ammonia in the blood (owing to diminished clearance of ammonia by the liver) underlie the pathogenesis of HE
-
Some degree of cerebral edema is observed in all grades of HE
-
The occurrence of any neuropsychiatric manifestation in patients with liver disease should be treated as HE unless proven otherwise
-
An acute episode of HE is managed by a tripartite strategy: ruling out other causes of encephalopathy, identifying the precipitating cause and initiating empiric therapy
-
Rifaximin and lactulose are the only two medications approved by FDA for long-term treatment of HE
-
Work-up for liver transplantation must be initiated as early as possible
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Ferenci, P. et al. Hepatic encephalopathy—definition, nomenclature, diagnosis, and quantification: final report of the working party at the 11th World Congresses of Gastroenterology, Vienna, 1998. Hepatology 35, 716–721 (2002).
Shawcross, D. L., Wright, G., Olde Damink, S. W. & Jalan, R. Role of ammonia and inflammation in minimal hepatic encephalopathy. Metab. Brain Dis. 22, 125–138 (2007).
Shawcross, D. L., Davies, N. A., Williams, R. & Jalan, R. Systemic inflammatory response exacerbates the neuropsychological effects of induced hyperammonemia in cirrhosis. J. Hepatol. 40, 247–254 (2004).
Norenberg, M. D., Jayakumar, A. R., Rama Rao, K. V. & Panickar, K. S. New concepts in the mechanism of ammonia-induced astrocyte swelling. Metab. Brain Dis. 22, 219–234 (2007).
Schliess, F., Görg, B. & Häussinger, D. Pathogenetic interplay between osmotic and oxidative stress: the hepatic encephalopathy paradigm. Biol. Chem. 387, 1363–1370 (2006).
Ahboucha, S. & Butterworth, R. F. The neurosteroid system: an emerging therapeutic target for hepatic encephalopathy. Metab. Brain Dis. 22, 291–308 (2007).
Qadri, A. M., Ogunwale, B. O. & Mullen, K. D. Can we ignore minimal hepatic encephalopathy any longer? Hepatology 45, 547–548 (2007).
Talwalkar, J. A. & Kamath, P. S. Influence of recent advances in medical management on clinical outcomes of cirrhosis. Mayo Clin. Proc. 80, 1501–1508 (2005).
Poordad, F. F. Review article: the burden of hepatic encephalopathy. Aliment. Pharmacol. Ther. 25 (Suppl. 1), 3–9 (2007).
Groeneweg, M. et al. Subclinical hepatic encephalopathy impairs daily functioning. Hepatology 28, 45–49 (1998).
Prasad, S. et al. Lactulose improves cognitive functions and health-related quality of life in patients with cirrhosis who have minimal hepatic encephalopathy. Hepatology 45, 549–559 (2007).
Bajaj, J. S. et al. Navigation skill impairment: Another dimension of the driving difficulties in minimal hepatic encephalopathy. Hepatology 47, 596–604 (2008).
Bajaj, J. S., Saeian, K., Hafeezullah, M., Hoffmann, R. G. & Hammeke, T. A. Patients with minimal hepatic encephalopathy have poor insight into their driving skills. Clin. Gastroenterol. Hepatol. 6, 1135–1139 (2008).
Bajaj, J. S. et al. The effect of fatigue on driving skills in patients with hepatic encephalopathy. Am. J. Gastroenterol. 104, 898–905 (2009).
Kircheis, G. et al. Hepatic encephalopathy and fitness to drive. Gastroenterology 137, 1706–1715.e9 (2009).
Bajaj, J. S. et al. Minimal hepatic encephalopathy is associated with motor vehicle crashes: the reality beyond the driving test. Hepatology 50, 1175–1183 (2009).
Bustamante, J. et al. Prognostic significance of hepatic encephalopathy in patients with cirrhosis. J. Hepatol. 30, 890–895 (1999).
Zieve, L. Pathogenesis of hepatic encephalopathy. Metab. Brain Dis. 2, 147–165 (1987).
Cooper, A. J. & Plum, F. Biochemistry and physiology of brain ammonia. Physiol. Rev. 67, 440–519 (1987).
Ytrebø, L. M. et al. Interorgan ammonia, glutamate, and glutamine trafficking in pigs with acute liver failure. Am. J. Physiol. Gastrointest. Liver Physiol. 291, G373–G381 (2006).
Olde Damink, S. W., Jalan, R. & Dejong, C. H. Interorgan ammonia trafficking in liver disease. Metab. Brain Dis. 24, 169–181 (2009).
Hahn, M., Massen, O., Nencki, M. & Pavlov, I. Die Eck'sche fistel zwischen der unteren hohlvene under pfortader und ihre folgen fur den organismus [German]. Arch. Exp. Pathol. Pharm. 32, 161–210 (1893).
Shawcross, D. L., Olde Damink, S. W., Butterworth, R. F. & Jalan, R. Ammonia and hepatic encephalopathy: the more things change, the more they remain the same. Metab. Brain Dis. 20, 169–179 (2005).
Phillips, G., Schwartz, R., Gabuzda, G. & Davidson, C. The syndrome of impending hepatic coma in patients with cirrhosis of the liver given certain nitrogenous substances. N. Engl. J. Med. 247, 239–246 (1952).
Lockwood, A. H., Yap, E. W. & Wong, W. H. Cerebral ammonia metabolism in patients with severe liver disease and minimal hepatic encephalopathy. J. Cereb. Blood Flow Metab. 11, 337–341 (1991).
Thomas, J. W., Banner, C., Whitman, J., Mullen, K. D. & Freese, E. Changes in glutamate-cycle enzyme mRNA levels in a rat model of hepatic encephalopathy. Metab. Brain Dis. 3, 81–90 (1988).
Häussinger, D., Kircheis, G., Fischer, R., Schiliess, F. & vom Dahl, S. Hepatic encephalopathy in chronic liver disease: a clinical manifestation of astrocyte swelling and low-grade cerebral edema? J. Hepatol. 32, 1035–1038 (2000).
Cooper, A. J., McDonald, J. M., Gelbard, A. S., Gledhill, R. F. & Duffy, T. E. The metabolic fate of 13N-labeled ammonia in rat brain. J. Biol. Chem. 254, 4982–4992 (1979).
Häussinger, D. & Schliess, F. Pathogenetic mechanisms of hepatic encephalopathy. Gut 57, 1156–1165 (2008).
Tanigami, H. et al. Effect of glutamine synthetase inhibition on astrocyte swelling and altered astroglial protein expression during hyperammonemia in rats. Neuroscience 131, 437–449 (2005).
Rose, C., Kresse, W. & Kettenmann, H. Acute insult of ammonia leads to calcium-dependent glutamate release from cultured astrocytes, an effect of pH. J. Biol. Chem. 280, 20937–20944 (2005).
Rose, C. Effect of ammonia on astrocytic glutamate uptake/release mechanisms. J. Neurochem. 97 (Suppl. 1), 11–15 (2006).
Hertz, L., Murthy, C. R., Lai, J. C., Fitzpatrick, S. M. & Cooper, A. J. Some metabolic effects of ammonia on astrocytes and neurons in primary cultures. Neurochem. Pathol. 6, 97–129 (1987).
Córdoba, J., Sanpedro, F., Alonso, J. & Rovira, A. 1H magnetic resonance in the study of hepatic encephalopathy in humans. Metab. Brain Dis. 17, 415–429 (2002).
Häussinger, D. Low grade cerebral edema and the pathogenesis of hepatic encephalopathy in cirrhosis. Hepatology 43, 1187–1190 (2006).
Rovira, A., Alonso, J. & Córdoba, J. MR imaging findings in hepatic encephalopathy. AJNR Am. J. Neuroradiol. 29, 1612–1621 (2008).
Shawcross, D. L. et al. Low myo-inositol and high glutamine levels in brain are associated with neuropsychological deterioration after induced hyperammonemia. Am. J. Physiol. Gastrointest. Liver Physiol. 287, G503–G509 (2004).
Butterworth, R. F. Pathophysiology of hepatic encephalopathy: The concept of synergism. Hepatol. Res. 38, S116–S121 (2008).
Gregorios, J. B., Mozes, L. W. & Norenberg, M. D. Morphologic effects of ammonia on primary astrocyte cultures. II. Electron microscopic studies. J. Neuropathol. Exp. Neurol. 44, 404–414 (1985).
Norenberg, M. D. The role of astrocytes in hepatic encephalopathy. Neurochem. Pathol. 6, 13–33 (1987).
Shawcross, D. & Jalan, R. The pathophysiologic basis of hepatic encephalopathy: central role for ammonia and inflammation. Cell. Mol. Life Sci. 62, 2295–2304 (2005).
Häussinger, D. & Schliess, F. Astrocyte swelling and protein tyrosine nitration in hepatic encephalopathy. Neurochem. Int. 47, 64–70 (2005).
Moldawer, L. L. et al. Cachectin/tumor necrosis factor-α alters red blood cell kinetics and induces anemia in vivo. FASEB J. 3, 1637–1643 (1989).
de Vries, H. E. et al. The influence of cytokines on the integrity of the blood-brain barrier in vitro. J. Neuroimmunol. 64, 37–43 (1996).
Didier, N. et al. Secretion of interleukin-1β by astrocytes mediates endothelin-1 and tumour necrosis factor-α effects on human brain microvascular endothelial cell permeability. J. Neurochem. 86, 246–254 (2003).
Duchini, A. Effects of tumor necrosis factor-α and interleukin-6 on fluid-phase permeability and ammonia diffusion in CNS-derived endothelial cells. J. Investig. Med. 44, 474–482 (1996).
Cagnin, A., Taylor-Robinson, S. D., Forton, D. M. & Banati, R. B. In vivo imaging of cerebral “peripheral benzodiazepine binding sites” in patients with hepatic encephalopathy. Gut 55, 547–553 (2006).
Ahboucha, S. & Butterworth, R. F. The neurosteroid system: implication in the pathophysiology of hepatic encephalopathy. Neurochem. Int. 52, 575–587 (2008).
Baulieu, E. E. Neurosteroids: a novel function of the brain. Psychoneuroendocrinology 23, 963–987 (1998).
Papadopoulos, V. et al. Translocator protein (18kDa): new nomenclature for the peripheral-type benzodiazepine receptor based on its structure and molecular function. Trends Pharmacol. Sci. 27, 402–409 (2006).
Papadopoulos, V., Lecanu, L., Brown, R. C., Han, Z. & Yao, Z. X. Peripheral-type benzodiazepine receptor in neurosteroid biosynthesis, neuropathology and neurological disorders. Neuroscience 138, 749–756 (2006).
Desjardins, P. & Butterworth, R. F. The “peripheral-type” benzodiazepine (omega 3) receptor in hyperammonemic disorders. Neurochem. Int. 41, 109–114 (2002).
Bélanger, M., Desjardins, P., Chatauret, N., Rose, C. & Butterworth, R. F. Mild hypothermia prevents brain edema and attenuates up-regulation of the astrocytic benzodiazepine receptor in experimental acute liver failure. J. Hepatol. 42, 694–699 (2005).
Ahboucha, S., Coyne, L., Hirakawa, R., Butterworth, R. F. & Halliwell, R. F. An interaction between benzodiazepines and neuroactive steroids at GABAA receptors in cultured hippocampal neurons. Neurochem. Int. 48, 703–707 (2006).
Murthy, C. R., Rama Rao, K. V., Bai, G. & Norenberg, M. D. Ammonia-induced production of free radicals in primary cultures of rat astrocytes. J. Neurosci. Res. 66, 282–298 (2001).
Hermenegildo, C., Monfort, P. & Felipo, V. Activation of N-methyl-D-aspartate receptors in rat brain in vivo following acute ammonia intoxication: characterization by in vivo brain microdialysis. Hepatology 31, 709–715 (2000).
Hilgier, W., Anderzhanova, E., Oja, S. S., Saransaari, P. & Albrecht, J. Taurine reduces ammonia- and N-methyl-D-aspartate-induced accumulation of cyclic GMP and hydroxyl radicals in microdialysates of the rat striatum. Eur. J. Pharmacol. 468, 21–25 (2003).
Reinehr, R. et al. Hypoosmotic swelling and ammonia increase oxidative stress by NADPH oxidase in cultured astrocytes and vital brain slices. Glia 55, 758–771 (2007).
Albrecht, J. & Norenberg, M. D. Glutamine: a Trojan horse in ammonia neurotoxicity. Hepatology 44, 788–794 (2006).
Schliess, F. et al. Ammonia induces MK-801-sensitive nitration and phosphorylation of protein tyrosine residues in rat astrocytes. FASEB J. 16, 739–741 (2002).
Mullen, K. D. & Jones, E. A. Natural benzodiazepines and hepatic encephalopathy. Semin. Liver Dis. 16, 255–264 (1996).
Mullen, K. D., Cole, M. & Foley, J. M. Neurological deficits in “awake” cirrhotic patients on hepatic encephalopathy treatment: missed metabolic or metal disorder? Gastroenterology 111, 256–257 (1996).
Rose, C. et al. Manganese deposition in basal ganglia structures results from both portal-systemic shunting and liver dysfunction. Gastroenterology 117, 640–644 (1999).
Naegele, T. et al. MR imaging and 1H spectroscopy of brain metabolites in hepatic encephalopathy: time-course of renormalization after liver transplantation. Radiology 216, 683–691 (2000).
Aggarwal, A. et al. Reversible Parkinsonism and T1W pallidal hyperintensities in acute liver failure. Mov. Disord. 21, 1986–1990 (2006).
Krieger, D. et al. Manganese and chronic hepatic encephalopathy. Lancet 346, 270–274 (1995).
Tsuji, H. et al. A case of liver cirrhosis associated with chronic subdural hematoma and hepatic encephalopathy [Japanese]. Fukuoka Igaku Zasshi. 82, 528–532 (1991).
Foreman, M. G., Mannino, D. M. & Moss, M. Cirrhosis as a risk factor for sepsis and death: analysis of the National Hospital Discharge Survey. Chest 124, 1016–1020 (2003).
Conn, H. O. et al. Comparison of lactulose and neomycin in the treatment of chronic portal-systemic encephalopathy. A double blind controlled trial. Gastroenterology 72, 573–583 (1977).
Bajaj, J. S., Wade, J. B. & Sanyal, A. J. Spectrum of neurocognitive impairment in cirrhosis: Implications for the assessment of hepatic encephalopathy. Hepatology 50, 2014–2021 (2009).
Parsons-Smith, B. G., Summerskill, W. H., Dawson, A. M. & Sherlock, S. The electroencephalograph in liver disease. Lancet 273, 867–871 (1957).
Jennett, B., Teasdale, G., Braakman, R., Minderhoud, J. & Knill-Jones, R. Predicting outcome in individual patients after severe head injury. Lancet 15, 1031–1034 (1976).
Montagnese, S., Amodio, P. & Morgan, M. Y. Methods for diagnosing hepatic encephalopathy in patients with cirrhosis: a multidimensional approach. Metab. Brain Dis. 19, 281–312 (2004).
Amodio, P., Montagnese, S., Gatta, A. & Morgan, M. Y. Characteristics of minimal hepatic encephalopathy. Metab. Brain Dis. 19, 253–267 (2004).
Hassanein, T. I., Hilsabeck, R. C. & Perry, W. Introduction to the Hepatic Encephalopathy Scoring Algorithm (HESA). Dig. Dis. Sci. 53, 529–538 (2008).
Weissenborn, K. et al. Neuropsychological characterization of hepatic encephalopathy. J. Hepatol. 34, 768–773 (2001).
Randolph, C. et al. Neuropsychological assessment of hepatic encephalopathy: ISHEN practice guidelines. Liver Int. 29, 629–635 (2009).
RED Hepatic Encephalopathy [Spanish]. Spanish normative data for hepatic encephalopathy [online], (2010).
Meyer, T., Eshelman, A. & Abouljoud, M. Neuropsychological changes in a large sample of liver transplant candidates. Transplant. Proc. 38, 3559–3560 (2006).
Sorrell, J. H., Zolnikov, B. J., Sharma, A. & Jinnai, I. Cognitive impairment in people diagnosed with end-stage liver disease evaluated for liver transplantation. Psychiatry Clin. Neurosci. 60, 174–181 (2006).
Bajaj, J. S. et al. Inhibitory control test is a simple method to diagnose minimal hepatic encephalopathy and predict development of overt hepatic encephalopathy. Am. J. Gastroenterol. 102, 754–760 (2007).
Bajaj, J. S. et al. Inhibitory control test for the diagnosis of minimal hepatic encephalopathy. Gastroenterology 135, 1591–1600 (2008).
Mardini, H., Saxby, B. K. & Record, C. O. Computerized psychometric testing in minimal encephalopathy and modulation by nitrogen challenge and liver transplant. Gastroenterology 135, 1582–1590 (2008).
Kircheis, G., Wettstein, M., Timmermann, L., Schnitzler, A. & Häussinger, D. Critical flicker frequency for quantification of low-grade hepatic encephalopathy. Hepatology 35, 357–366 (2002).
Kircheis, G. et al. Diagnostic and prognostic values of critical flicker frequency determination as new diagnostic tool for objective HE evaluation in patients undergoing TIPS implantation. Eur. J. Gastroenterol. Hepatol. 21, 1383–1394 (2009).
Amodio, P. et al. Electroencephalographic analysis for the assessment of hepatic encephalopathy: comparison of non-parametric and parametric spectral estimation techniques. Neurophysiol. Clin. 39, 107–115 (2009).
Ong, J. P. et al. Correlation between ammonia levels and the severity of hepatic encephalopathy. Am. J. Med. 114, 188–193 (2003).
Morgan, M. Y., Alonso, M. & Stanger, L. C. Lactitol and lactulose for the treatment of subclinical hepatic encephalopathy in cirrhotic patients. A randomised, cross-over study. J. Hepatol. 8, 208–217 (1989).
Morgan, M. Y. & Hawley, K. E. Lactitol vs. lactulose in the treatment of acute hepatic encephalopathy in cirrhotic patients: a double-blind, randomized trial. Hepatology 7, 1278–1284 (1987).
Morgan, M. Y., Hawley, K. E. & Stambuk, D. Lactitol versus lactulose in the treatment of chronic hepatic encephalopathy. A double-blind, randomised, cross-over study. J. Hepatol. 4, 236–244 (1987).
van Leeuwen, P. A., van Berlo, C. L. & Soeters, P. B. New mode of action for lactulose. Lancet 1, 55–56 (1988).
Bajaj, J. S. Management options for minimal hepatic encephalopathy. Expert Rev. Gastroenterol. Hepatol. 2, 785–790 (2008).
Mullen, K. D., Amodio, P & Morgan, M. Y. Therapeutic studies in hepatic encephalopathy. Metab. Brain Dis. 22, 407–423 (2007).
Bass, N. M. et al. Rifaximin treatment in hepatic encephalopathy. N. Engl. J. Med. 362, 1071–1081 (2010).
Plauth, M. et al. ESPEN Guidelines on Enteral Nutrition: Liver disease. Clin. Nutr. 25, 285–294 (2006).
Mullen, K. D. & Dasarathy, S. Protein restriction in hepatic encephalopathy: necessary evil or illogical dogma? J. Hepatol. 41, 147–148 (2004).
Muto, Y. et al. Effects of oral branched-chain amino acid granules on event-free survival in patients with liver cirrhosis. Clin. Gastroenterol. Hepatol. 3, 705–713 (2005).
Als-Nielsen, B., Koretz, R. L., Kjaergard, L. L. & Gluud, C. Branched-chain amino acids for hepatic encephalopathy. Cochrane Database of Systematic Reviews, Issue 1. Art. No.: CD001939. doi:10.1002/14651858.CD001939 (2003).
Amodio, P. et al. Vegetarian diets in hepatic encephalopathy: facts or fantasies? Dig. Liver Dis. 33, 492–500 (2001).
Bajaj, J. S. Review article: the modern management of hepatic encephalopathy. Aliment. Pharmacol. Ther. 31, 537–547 (2010).
Stewart, C. A., Malinchoc, M., Kim, W. R. & Kamath, P. S. Hepatic encephalopathy as a predictor of survival in patients with end-stage liver disease. Liver Transpl. 13, 1366–1371 (2007).
Fanelli, F. et al. Management of refractory hepatic encephalopathy after insertion of TIPS: long-term results of shunt reduction with hourglass-shaped balloon-expandable stent-graft. AJR Am. J. Roentgenol. 193, 1696–1702 (2009).
Montoliu, C. et al. Activation of soluble guanylate cyclase by nitric oxide in lymphocytes correlates with minimal hepatic encephalopathy in cirrhotic patients. J. Mol. Med. 85, 237–245 (2007).
Albrecht, J. Cyclic GMP in blood and minimal hepatic encephalopathy: fine-tuning of the diagnosis. J. Mol. Med. 85, 203–205 (2007).
Marchesini, G., Fabbri, A., Bianchi, G., Brizi, M. & Zoli, M. Zinc supplementation and amino acid-nitrogen metabolism in patients with advanced cirrhosis. Hepatology 23, 1084–1092 (1996).
Bresci, G., Parisi, G. & Banti, S. Management of hepatic encephalopathy with oral zinc supplementation: a long-term treatment. Eur. J. Med. 2, 414–416 (1993).
Morgan, M. Y., Jakobovits, A. W., James, I. M. & Sherlock, S. Successful use of bromocriptine in the treatment of chronic hepatic encephalopathy. Gastroenterology 78, 663–670 (1980).
Sushma, S. et al. Sodium benzoate in the treatment of acute hepatic encephalopathy: a double-blind randomized trial. Hepatology 16, 138–144 (1992).
Kircheis, G. et al. Therapeutic efficacy of L-ornithine-L-aspartate infusions in patients with cirrhosis and hepatic encephalopathy: results of a placebo-controlled, double-blind study. Hepatology 25, 1351–1360 (1997).
Kircheis, G., Wettstein, M., Dahl, S. & Häussinger, D. Clinical efficacy of L-ornithine-L-aspartate in the management of hepatic encephalopathy. Metab. Brain Dis. 17, 453–462 (2002).
Acknowledgements
Désirée Lie, University of California, Irvine, CA, is the author of and is solely responsible for the content of the learning objectives, questions and answers of the MedscapeCME-accredited continuing medical education activity associated with this article.
Author information
Authors and Affiliations
Contributions
R. Prakash and K. D. Mullen contributed equally to researching data for the article, to substantial discussion of the content, to writing the article, and to reviewing and/or editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
K. D. Mullen is a consultant for Ocera Therapeutics and a consultant and speaker for Salix Pharmaceuticals (he has received honoria, but not for this manuscript). R. Prakash declares no competing interests.
Rights and permissions
About this article
Cite this article
Prakash, R., Mullen, K. Mechanisms, diagnosis and management of hepatic encephalopathy. Nat Rev Gastroenterol Hepatol 7, 515–525 (2010). https://doi.org/10.1038/nrgastro.2010.116
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrgastro.2010.116
This article is cited by
-
Confusion assessment method accurately screens for hepatic encephalopathy and predicts short-term mortality in hospitalized patients with cirrhosis
Metabolic Brain Disease (2023)
-
J-difference GABA-edited MRS reveals altered cerebello-thalamo-cortical metabolism in patients with hepatic encephalopathy
Metabolic Brain Disease (2023)
-
Ameliorative effect of oregano (Origanum vulgare) versus silymarin in experimentally induced hepatic encephalopathy
Scientific Reports (2022)
-
Mitochondrial Changes in Rat Brain Endothelial Cells Associated with Hepatic Encephalopathy: Relation to the Blood–Brain Barrier Dysfunction
Neurochemical Research (2022)
-
Therapeutic potential of probiotics – Lactobacillus plantarum UBLP40 and Bacillus clausii UBBC07 on thioacetamide-induced acute hepatic encephalopathy in rats
Metabolic Brain Disease (2022)