Key Points
-
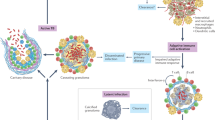
Mycobacterium tuberculosis survives in the host in antigen-presenting cells (APCs) such as macrophages and dendritic cells.
-
APCs present antigens in association with major histocompatibility complex (MHC) class II molecules to stimulate CD4+ T cells, and this process is essential for containing M. tuberculosis infection.
-
Immune evasion allows M. tuberculosis to establish persistent or latent infection in APCs.
-
M. tuberculosis infection of macrophages results in Toll-like receptor 2 (TLR2)-dependent inhibition of MHC class II transactivator (CIITA) and MHC class II molecule expression and of MHC class II antigen presentation, providing a mechanism for immune evasion.
-
The TLR2-dependent reduction of antigen presentation might reflect a general mechanism of negative-feedback regulation that prevents excessive T cell-mediated inflammation and that M. tuberculosis has subverted for the purposes of immune evasion.
-
Inhibition of antigen presentation creates a niche for M. tuberculosis survival in infected APCs and for its evasion of recognition by CD4+ T cells.
Abstract
Mycobacterium tuberculosis survives in antigen-presenting cells (APCs) such as macrophages and dendritic cells. APCs present antigens in association with major histocompatibility complex (MHC) class II molecules to stimulate CD4+ T cells, and this process is essential to contain M. tuberculosis infection. Immune evasion allows M. tuberculosis to establish persistent or latent infection in macrophages and results in Toll-like receptor 2 (TLR2)-dependent inhibition of MHC class II transactivator expression, MHC class II molecule expression and antigen presentation. This reduction of antigen presentation might reflect a general mechanism of negative-feedback regulation that prevents excessive T cell-mediated inflammation and that M. tuberculosis has subverted to create a niche for survival in infected macrophages and evasion of recognition by CD4+ T cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Barnes, P. F., Bloch, A. B., Davidson, P. T. & Snider, D. E. Jr. Tuberculosis in patients with human immunodeficiency virus infection. N. Engl. J. Med. 324, 1644–1650 (1991).
Ladel, C. H., Daugelat, S. & Kaufmann, S. H. E. Immune response to Mycobacterium bovis bacille Calmette Guérin infection in major histocompatibility complex class I- and II-deficient knock-out mice: contribution of CD4 and CD8 T cells to acquired resistance. Eur. J. Immunol. 25, 377–384 (1995).
Mogues, T., Goodrich, M. E., Ryan, L., LaCourse, R. & North, R. J. The relative importance of T cell subsets in immunity and immunopathology of airborne Mycobacterium tuberculosis infection in mice. J. Exp. Med. 193, 271–280 (2001).
Caruso, A. M. et al. Mice deficient in CD4 T cells have only transiently diminished levels of IFN-γ, yet succumb to tuberculosis. J. Immunol. 162, 5407–5416 (1999).
Scanga, C. A. et al. Depletion of CD4+ T cells causes reactivation of murine persistent tuberculosis despite continued expression of interferon γ and nitric oxide synthase 2. J. Exp. Med. 192, 347–358 (2000).
Repique, C. J. et al. Susceptibility of mice deficient in the MHC class II transactivator to infection with Mycobacterium tuberculosis. Scand. J. Immunol. 58, 15–22 (2003).
Gallegos, A. M., Pamer, E. G. & Glickman, M. S. Delayed protection by ESAT-6-specific effector CD4+ T cells after airborne M. tuberculosis infection. J. Exp. Med. 205, 2359–2368 (2008). This work uses transgenic mice expressing a T cell receptor specific for the M. tuberculosis antigen early secretory antigenic target 6 (ESAT6) along with adoptive transfer models to demonstrate the activation of M. tuberculosis -specific T cells in pulmonary lymph nodes and their subsequent trafficking to the lung.
Wolf, A. J. et al. Initiation of the adaptive immune response to Mycobacterium tuberculosis depends on antigen production in the local lymph node, not the lungs. J. Exp. Med. 205, 105–115 (2008). This study uses transgenic mice expressing a T cell receptor specific for a M. tuberculosis antigen 85B to show the initial activation of M. tuberculosis -specific T cells in lung-draining lymph nodes and to define a lag period before antigen is presented to launch T cell responses.
Russell, M. S. et al. A reduced antigen load in vivo, rather than weak inflammation, causes a substantial delay in CD8+ T cell priming against Mycobacterium bovis (bacillus Calmette-Guerin). J. Immunol. 179, 211–220 (2007).
Ferwerda, G. et al. NOD2 and Toll-like receptors are nonredundant recognition systems of Mycobacterium tuberculosis. PLoS Pathog. 1, e34 (2005). This article compares the roles of NOD2 and TLR2 in responses to M. tuberculosis .
Geijtenbeek, T. B. et al. Mycobacteria target DC-SIGN to suppress dendritic cell function. J. Exp. Med. 197, 7–17 (2003). An investigation that defines an important role for DC-SIGN as a receptor for M. tuberculosis .
Flynn, J. L. et al. An essential role for interferon γ in resistance to Mycobacterium tuberculosis infection. J. Exp. Med. 178, 2249–2254 (1993).
Dalton, D. K. et al. Multiple defects of immune cell function in mice with disrupted interferon-gamma genes. Science 259, 1739–1742 (1993).
Cooper, A. M. et al. Disseminated tuberculosis in interferon γ gene-disrupted mice. J. Exp. Med. 178, 2243–2247 (1993). References 12, 13 and 14 describe the use of IFNγ-deficient mice to demonstrate the key role of IFNγ in resistance to M. tuberculosis infection.
Dupuis, S. et al. Impairment of mycobacterial but not viral immunity by a germline human STAT1 mutation. Science 293, 300–303 (2001).
MacMicking, J. D., Taylor, G. A. & McKinney, J. D. Immune control of tuberculosis by IFN-γ-inducible LRG-47. Science 302, 654–659 (2003).
Ottenhoff, T. H., Kumararatne, D. & Casanova, J. L. Novel human immunodeficiencies reveal the essential role of type-I cytokines in immunity to intracellular bacteria. Immunol. Today 19, 491–494 (1998). A review of the seminal publications on the essential role of IFNγ in human resistance to mycobacterial infection.
Banaiee, N., Kincaid, E. Z., Buchwald, U., Jacobs, W. R. Jr. & Ernst, J. D. Potent inhibition of macrophage responses to IFN-γ by live virulent Mycobacterium tuberculosis is independent of mature mycobacterial lipoproteins but dependent on TLR2. J. Immunol. 176, 3019–3027 (2006).
Fortune, S. M. et al. Mycobacterium tuberculosis inhibits macrophage responses to IFN-γ through myeloid differentiation factor 88-dependent and -independent mechanisms. J. Immunol. 172, 6272–6280 (2004).
Kincaid, E. Z. & Ernst, J. D. Mycobacterium tuberculosis exerts gene-selective inhibition of transcriptional responses to IFN-γ without inhibiting STAT1 function. J. Immunol. 171, 2042–2049 (2003).
Kincaid, E. Z. et al. Codominance of TLR2-dependent and TLR2-independent modulation of MHC class II in Mycobacterium tuberculosis infection in vivo. J. Immunol. 179, 3187–3195 (2007).
Ting, L. M., Kim, A. C., Cattamanchi, A. & Ernst, J. D. Mycobacterium tuberculosis inhibits IFN-γ transcriptional responses without inhibiting activation of STAT1. J. Immunol. 163, 3898–3906 (1999). References 20 and 22 report that M. tuberculosis inhibits IFNγ-stimulated induction of certain genes, and the authors dissect the signalling mechanisms that are involved.
Pai, R. K., Convery, M., Hamilton, T. A., Boom, W. H. & Harding, C. V. Inhibition of IFN-γ-induced class II transactivator expression by a 19-kDa lipoprotein from Mycobacterium tuberculosis: a potential mechanism for immune evasion. J. Immunol. 171, 175–184 (2003). This work defines a mechanism for the inhibition of MHC class II molecule expression that is mediated by M. tuberculosis or M. tuberculosis lipoprotein through the TLR2-dependent inhibition of CIITA induction by IFNγ.
Pai, R. K. et al. Prolonged Toll-like receptor signaling by Mycobacterium tuberculosis and its 19-kilodalton lipoprotein inhibits gamma interferon-induced regulation of selected genes in macrophages. Infect. Immun. 72, 6603–6614 (2004).
Pennini, M. E. et al. CCAAT/enhancer-binding protein β and δ binding to CIITA promoters is associated with the inhibition of CIITA expression in response to Mycobacterium tuberculosis 19-kDa lipoprotein. J. Immunol. 179, 6910–6918 (2007). This paper reports a role for C/EBPβ and C/EBPδ in transcriptional control of CIITA through their binding to CIITA promoters and shows that this mechanism is induced in response to lipoprotein TLR2 agonists from M. tuberculosis .
Pennini, M. E., Pai, R. K., Schultz, D. C., Boom, W. H. & Harding, C. V. Mycobacterium tuberculosis 19-kDa lipoprotein inhibits IFN-γ-induced chromatin remodeling of MHC2TA by TLR2 and MAPK signaling. J. Immunol. 176, 4323–4330 (2006).
Lafuse, W. P., Alvarez, G. R., Curry, H. M. & Zwilling, B. S. Mycobacterium tuberculosis and Mycobacterium avium inhibit IFN-γ-induced gene expression by TLR2-dependent and independent pathways. J. Interferon Cytokine Res. 26, 548–561 (2006).
Wang, Y., Curry, H. M., Zwilling, B. S. & Lafuse, W. P. Mycobacteria inhibition of IFN-γ induced HLA-DR gene expression by up-regulating histone deacetylation at the promoter region in human THP-1 monocytic cells. J. Immunol. 174, 5687–5694 (2005).
Vazquez, N., Greenwell-Wild, T., Rekka, S., Orenstein, J. M. & Wahl, S. M. Mycobacterium avium-induced SOCS contributes to resistance to IFN-γ-mediated mycobactericidal activity in human macrophages. J. Leukoc. Biol. 80, 1136–1144 (2006).
Hussain, S., Zwilling, B. S. & Lafuse, W. P. Mycobacterium avium infection of mouse macrophages inhibits IFN-γ Janus kinase-STAT signaling and gene induction by down-regulation of the IFN-γ receptor. J. Immunol. 163, 2041–2048 (1999).
Ehrt, S. et al. Reprogramming of the macrophage transcriptome in response to interferon-γ and Mycobacterium tuberculosis: signaling roles of nitric oxide synthase-2 and phagocyte oxidase. J. Exp. Med. 194, 1123–1140 (2001).
Noss, E. H. et al. Toll-like receptor 2-dependent inhibition of macrophage class II MHC expression and antigen processing by 19 kD lipoprotein of Mycobacterium tuberculosis. J. Immunol. 167, 910–918 (2001). This work shows the ability of M. tuberculosis and M. tuberculosis lipoprotein LpqH to use TLR2 dependent mechanisms to inhibit IFNγ-induced expression of MHC class II molecules, suggesting a mechanism for immune evasion.
Gercken, J., Pryjma, J., Ernst, M. & Flad, H. D. Defective antigen presentation by Mycobacterium tuberculosis infected monocytes. Infect. Immun. 62, 3472–3478 (1994).
Hmama, Z., Gabathuler, R., Jefferies, W. A., de Jong, G. & Reiner, N. E. Attenuation of HLA-DR expression by mononuclear phagocytes infected with Mycobacterium tuberculosis is related to intracellular sequestration of immature class II heterodimers. J. Immunol. 161, 4882–4893 (1998).
Noss, E. H., Harding, C. V. & Boom, W. H. Mycobacterium tuberculosis inhibits MHC class II antigen processing in murine bone marrow macrophages. Cell. Immunol. 201, 63–74 (2000).
Pancholi, P., Mirza, A., Bhardwaj, N. & Steinman, R. M. Sequestration from immune CD4+ T cells of mycobacteria growing in human macrophages. Science 260, 984–986 (1993).
VanHeyningen, T. K., Collins, H. L. & Russell, D. G. IL-6 produced by macrophages infected with Mycobacterium species suppresses T cell responses. J. Immunol. 158, 330–337 (1997).
Wojciechowski, W., DeSanctis, J., Skamene, E. & Radzioch, D. Attenuation of MHC class II expression in macrophages infected with Mycobacterium bovis bacillus Calmette-Guerin involves class II transactivator and depends on the Nramp1 gene. J. Immunol. 163, 2688–2696 (1999).
De Lerma Barbaro, A. et al. Distinct regulation of HLA class II and class I cell surface expression in the THP-1 macrophage cell line after bacterial phagocytosis. Eur. J. Immunol. 29, 499–511 (1999).
Gehring, A. J., Dobos, K. M., Belisle, J. T., Harding, C. V. & Boom, W. H. Mycobacterium tuberculosis LprG (Rv1411c): a novel TLR-2 ligand that inhibits human macrophage class II MHC antigen processing. J. Immunol. 173, 2660–2668 (2004).
Pecora, N. D., Gehring, A. J., Canaday, D. H., Boom, W. H. & Harding, C. V. Mycobacterium tuberculosis LprA is a lipoprotein agonist of TLR2 that regulates innate immunity and APC function. J. Immunol. 177, 422–429 (2006).
Thoma-Uszynski, S. et al. Induction of direct antimicrobial activity through mammalian Toll-like receptors. Science 291, 1544–1547 (2001). This study demonstrates a role for TLR2 in responses to mycobacteria and mycobacterial lipoproteins, including the induction of antimicrobial responses.
Jung, S. B. et al. The mycobacterial 38-kilodalton glycolipoprotein antigen activates the mitogen-activated protein kinase pathway and release of proinflammatory cytokines through Toll-like receptors 2 and 4 in human monocytes. Infect. Immun. 74, 2686–2696 (2006).
Jin, M. S. et al. Crystal structure of the TLR1-TLR2 heterodimer induced by binding of a tri-acylated lipopeptide. Cell 130, 1071–1082 (2007). This work characterizes the ligand-dependent assembly of TLR2–TLR1 heterodimers and the crystal structure of the receptor–ligand complex.
Tapping, R. I. & Tobias, P. S. Mycobacterial lipoarabinomannan mediates physical interactions between TLR1 and TLR2 to induce signaling. J. Endotoxin Res. 9, 264–268 (2003).
Takeuchi, O. et al. Cutting edge: role of Toll-like receptor 1 in mediating immune response to microbial lipoproteins. J. Immunol. 169, 10–14 (2002).
Ozinsky, A. et al. The repertoire for pattern recognition of pathogens by the innate immune system is defined by cooperation between Toll-like receptors. Proc. Natl Acad. Sci. USA 97, 13766–13771 (2000).
Wyllie, D. H. et al. Evidence for an accessory protein function for Toll-like receptor 1 in anti-bacterial responses. J. Immunol. 165, 7125–7132 (2000).
Rezwan, M., Grau, T., Tschumi, A. & Sander, P. Lipoprotein synthesis in mycobacteria. Microbiology 153, 652–658 (2007).
Sankaran, K. & Wu, H. C. Lipid modification of bacterial prolipoprotein. Transfer of diacylglyceryl moiety from phosphatidylglycerol. J. Biol. Chem. 269, 19701–19706 (1994).
Sander, P. et al. Lipoprotein processing is required for virulence of Mycobacterium tuberculosis. Mol. Microbiol. 52, 1543–1552 (2004).
Hussain, M., Ichihara, S. & Mizushima, S. Mechanism of signal peptide cleavage in the biosynthesis of the major lipoprotein of the Escherichia coli outer membrane. J. Biol. Chem. 257, 5177–5182 (1982).
Sutcliffe, I. C. & Harrington, D. J. Lipoproteins of Mycobacterium tuberculosis: an abundant and functionally diverse class of cell envelope components. FEMS Microbiol. Rev. 28, 645–659 (2004).
Means, T. K. et al. The CD14 ligands lipoarabinomannan and lipopolysaccharide differ in their requirement for Toll-like receptors. J. Immunol. 163, 6748–6755 (1999).
Means, T. K. et al. Human Toll-like receptors mediate cellular activation by Mycobacterium tuberculosis. J. Immunol. 163, 3920–3927 (1999).
Underhill, D. M., Ozinsky, A., Smith, K. D. & Aderem, A. Toll-like receptor-2 mediates mycobacteria-induced proinflammatory signaling in macrophages. Proc. Natl Acad. Sci. USA 96, 14459–14463 (1999). This study demonstrates the role of TLR2 in recognition of M. tuberculosis .
Jones, B. W. et al. Different Toll-like receptor agonists induce distinct macrophage responses. J. Leukoc. Biol. 69, 1036–1044 (2001).
Pitarque, S. et al. The immunomodulatory lipoglycans, lipoarabinomannan and lipomannan, are exposed at the mycobacterial cell surface. Tuberculosis (Edinb.) 88, 560–565 (2008).
Nigou, J. et al. Mannan chain length controls lipoglycans signaling via and binding to TLR2. J. Immunol. 180, 6696–6702 (2008).
Gilleron, M., Nigou, J., Nicolle, D., Quesniaux, V. & Puzo, G. The acylation state of mycobacterial lipomannans modulates innate immunity response through toll-like receptor 2. Chem. Biol. 13, 39–47 (2006).
Gilleron, M., Bala, L., Brando, T., Vercellone, A. & Puzo, G. Mycobacterium tuberculosis H37Rv parietal and cellular lipoarabinomannans. Characterization of the acyl- and glyco-forms. J. Biol. Chem. 275, 677–684 (2000).
Briken, V., Porcelli, S. A., Besra, G. S. & Kremer, L. Mycobacterial lipoarabinomannan and related lipoglycans: from biogenesis to modulation of the immune response. Mol. Microbiol. 53, 391–403 (2004).
Hsu, F. F., Turk, J., Owens, R. M., Rhoades, E. R. & Russell, D. G. Structural characterization of phosphatidyl-myo-inositol mannosides from Mycobacterium bovis Bacillus Calmette Guerin by multiple-stage quadrupole ion-trap mass spectrometry with electrospray ionization. I. PIMs and lyso-PIMs. J. Am. Soc. Mass Spectrom. 18, 466–478 (2007).
Sandor, F. et al. Importance of extra- and intracellular domains of TLR1 and TLR2 in NFκB signaling. J. Cell Biol. 162, 1099–1110 (2003).
Dao, D. N. et al. Mycobacterium tuberculosis lipomannan induces apoptosis and interleukin-12 production in macrophages. Infect. Immun. 72, 2067–2074 (2004).
Nigou, J. et al. Mycobacterial lipoarabinomannans: modulators of dendritic cell function and the apoptotic response. Microbes Infect. 4, 945–953 (2002).
Bowdish, D. M. et al. MARCO, TLR2, and CD14 are required for macrophage cytokine responses to mycobacterial trehalose dimycolate and Mycobacterium tuberculosis. PLoS Pathog. 5, e1000474 (2009). This investigation shows signalling of trehalose dimycolate through TLR2 and the roles of CD14 and MARCO as accessory receptors.
Manicassamy, S. & Pulendran, B. Modulation of adaptive immunity with Toll-like receptors. Semin. Immunol. 21, 185–193 (2009).
Chu, R. S. et al. CpG oligodeoxynucleotides down-regulate macrophage class II MHC antigen processing. J. Immunol. 163, 1188–1194 (1999).
Steeg, P. S., Johnson, H. M. & Oppenheim, J. J. Regulation of murine macrophage Ia antigen expression by an immune interferon-like lymphokine: inhibitory effect of endotoxin. J. Immunol. 129, 2402–2406 (1982).
Sicher, S. C., Vazquez, M. A. & Lu, C. Y. Inhibition of macrophage Ia expression by nitric oxide. J. Immunol. 153, 1293–1300 (1994).
Koerner, T. J., Hamilton, T. A. & Adams, D. O. Suppressed expression of surface Ia on macrophages by lipopolysaccaride: evidence for regulation at the level of accumulation of mRNA. J. Immunol. 139, 239–243 (1987).
Underhill, D. M. et al. The Toll-like receptor 2 is recruited to macrophage phagosomes and discriminates between pathogens. Nature 401, 811–815 (1999). A study showing the recruitment of TLR2 to phagosomes, which allows localization of TLR2 at the site of expression of TLR2 agonists from microorganisms.
Neyrolles, O. et al. Lipoprotein access to MHC class I presentation during infection of murine macrophages with live mycobacteria. J. Immunol. 166, 447–457 (2001).
Beatty, W. L. et al. Trafficking and release of mycobacterial lipids from infected macrophages. Traffic 1, 235–247 (2000).
Beatty, W. L. & Russell, D. G. Identification of mycobacterial surface proteins released into subcellular compartments of infected macrophages. Infect. Immun. 68, 6997–7002 (2000).
Brightbill, H. D. et al. Host defense mechanisms triggered by microbial lipoproteins through Toll-like receptors. Science 285, 732–736 (1999). This work demonstrates that TLR2 recognizes lipoproteins from M. tuberculosis , including LpqH.
Stenger, S. & Modlin, R. L. Control of Mycobacterium tuberculosis through mammalian Toll-like receptors. Curr. Opin. Immunol. 14, 452–457 (2002).
Schorey, J. S. & Bhatnagar, S. Exosome function: from tumor immunology to pathogen biology. Traffic 9, 871–881 (2008).
Rhoades, E. et al. Identification and macrophage-activating activity of glycolipids released from intracellular Mycobacterium bovis BCG. Mol. Microbiol. 48, 875–888 (2003).
Reiling, N. et al. Cutting edge: Toll-like receptor (TLR) 2- and TLR4-mediated pathogen recognition in resistance to airborne infection with Mycobacterium tuberculosis. J. Immunol. 169, 3480–3484 (2002).
Drennan, M. B. et al. Toll-like receptor 2-deficient mice succumb to Mycobacterium tuberculosis infection. Am. J. Pathol. 164, 49–57 (2004).
Heldwein, K. A. et al. TLR2 and TLR4 serve distinct roles in the host immune response against Mycobacterium bovis BCG. J. Leukoc. Biol. 74, 277–286 (2003).
Sugawara, I. et al. Mycobacterial infection in TLR2 and TLR6 knockout mice. Microbiol. Immunol. 47, 327–336 (2003).
Abel, B. et al. Toll-like receptor 4 expression is required to control chronic Mycobacterium tuberculosis infection in mice. J. Immunol. 169, 3155–3162 (2002).
Bafica, A. et al. TLR9 regulates Th1 responses and cooperates with TLR2 in mediating optimal resistance to Mycobacterium tuberculosis. J. Exp. Med. 202, 1715–1724 (2005). This article is one of the many reports on the role of TLRs in resistance to M. tuberculosis . This study nicely shows the roles of TLR2 and TLR9 and finds that mice deficient for both TLR2 and TLR9 are more susceptible to M. tuberculosis infection than mice with a single deficiency in either receptor.
Fremond, C. M. et al. IL-1 receptor-mediated signal is an essential component of MyD88-dependent innate response to Mycobacterium tuberculosis infection. J. Immunol. 179, 1178–1189 (2007).
Fremond, C. M. et al. Fatal Mycobacterium tuberculosis infection despite adaptive immune response in the absence of MyD88. J. Clin. Invest. 114, 1790–1799 (2004).
Feng, C. G. et al. Mice lacking myeloid differentiation factor 88 display profound defects in host resistance and immune responses to Mycobacterium avium infection not exhibited by Toll-like receptor 2 (TLR2)- and TLR4-deficient animals. J. Immunol. 171, 4758–4764 (2003).
von Bernuth, H. et al. Pyogenic bacterial infections in humans with MyD88 deficiency. Science 321, 691–696 (2008).
Kamath, A. B., Alt, J., Debbabi, H. & Behar, S. M. Toll-like receptor 4-defective C3H/HeJ mice are not more susceptible than other C3H substrains to infection with Mycobacterium tuberculosis. Infect. Immun. 71, 4112–4118 (2003).
Holscher, C. et al. Containment of aerogenic Mycobacterium tuberculosis infection in mice does not require MyD88 adaptor function for TLR2, -4 and -9. Eur. J. Immunol. 38, 680–694 (2008).
Ben-Ali, M., Barbouche, M. R., Bousnina, S., Chabbou, A. & Dellagi, K. Toll-like receptor 2 Arg677Trp polymorphism is associated with susceptibility to tuberculosis in Tunisian patients. Clin. Diagn. Lab. Immunol. 11, 625–626 (2004).
Bochud, P. Y., Hawn, T. R. & Aderem, A. Cutting edge: a Toll-like receptor 2 polymorphism that is associated with lepromatous leprosy is unable to mediate mycobacterial signaling. J. Immunol. 170, 3451–3454 (2003).
Ogus, A. C. et al. The Arg753GLn polymorphism of the human Toll-like receptor 2 gene in tuberculosis disease. Eur. Respir. J. 23, 219–223 (2004).
Reith, W. & Boss, J. M. New dimensions of CIITA. Nature Immunol. 9, 713–714 (2008).
Reith, W. & Mach, B. The bare lymphocyte syndrome and the regulation of MHC expression. Annu. Rev. Immunol. 19, 331–373 (2001).
Masternak, K. et al. CIITA is a transcriptional coactivator that is recruited to MHC class II promoters by multiple synergistic interactions with an enhanceosome complex. Genes Dev. 14, 1156–1166 (2000).
Steimle, V., Siegrist, C. A., Mottet, A., Lisowska-Grospierre, B. & Mach, B. Regulation of MHC class II expression by interferon-gamma mediated by the transactivator gene CIITA. Science 265, 106–109 (1994).
Steimle, V., Otten, L. A., Zufferey, M. & Mach, B. Complementation cloning of an MHC class II transactivator mutated in hereditary MHC class II deficiency (or bare lymphocyte syndrome). Cell 75, 135–146 (1993). References 99 and 100 are seminal reports of the key role of CIITA in transcriptional regulation of MHC class II genes.
Harton, J. A. & Ting, J. P. Class II transactivator: mastering the art of major histocompatibility complex expression. Mol. Cell Biol. 20, 6185–6194 (2000).
Boss, J. M. & Jensen, P. E. Transcriptional regulation of the MHC class II antigen presentation pathway. Curr. Opin. Immunol. 15, 105–111 (2003).
Chang, C.H. & Flavell, R. A. Class II transactivator regulates the expression of multiple genes involved in antigen presentation. J. Exp. Med. 181, 765–767 (1995). This work uses transfection of cells to express CIITA and demonstrate that CIITA regulates not only MHC class II molecules but also other molecules involved in the MHC class II antigen processing and presentation pathway.
Chang, C. H., Guerder, G., Hong, S. C., van Ewijk, W. & Flavell, R. A. Mice lacking the MHC class II transactivator (CIITA) show tissue-specific impairment of MHC class II expression. Immunity 4, 167–178 (1996).
Muhlethaler-Mottet, A., Otten, L. A., Steimle, V. & Mach, B. Expression of MHC class II molecules in different cellular and functional compartments is controlled by differential usage of multiple promoters of the transactivator CIITA. EMBO J. 16, 2851–2860 (1997).
Pai, R. K., Askew, D., Boom, W. H. & Harding, C. V. Regulation of class II MHC expression in APCs: roles of types, I, III, and IV class II transactivator. J. Immunol. 169, 1326–1333 (2002).
Kielar, M. L., Sicher, S. C., Penfield, J. G., Jeyarajah, D. R. & Lu, C. Y. Nitric oxide inhibits INFγ-induced increases in CIITA mRNA abundance and activation of CIITA dependent genes–class II MHC, Ii and H-2M. Inflammation 24, 431–445 (2000).
Jang, S., Uematsu, S., Akira, S. & Salgame, P. IL-6 and IL-10 induction from dendritic cells in response to Mycobacterium tuberculosis is predominantly dependent on TLR2-mediated recognition. J. Immunol. 173, 3392–3397 (2004).
Nagabhushanam, V. et al. Innate inhibition of adaptive immunity: Mycobacterium tuberculosis-induced IL-6 inhibits macrophage responses to IFN-γ. J. Immunol. 171, 4750–4757 (2003).
Wolf, A. J. et al. Mycobacterium tuberculosis infects dendritic cells with high frequency and impairs their function in vivo. J. Immunol. 179, 2509–2519 (2007).
Gehring, A. J. et al. The Mycobacterium tuberculosis 19-kilodalton lipoprotein inhibits gamma interferon-regulated HLA-DR and FcγR1 on human macrophages through Toll-like receptor 2. Infect. Immun. 71, 4487–4497 (2003).
Ramachandra, L., Noss, E., Boom, W. H. & Harding, C. V. Processing of Mycobacterium tuberculosis antigen 85B involves intraphagosomal formation of peptide-major histocompatibility complex II complexes and is inhibited by live bacilli that decrease phagosome maturation. J. Exp. Med. 194, 1421–1432 (2001). This study uses subcellular fractionation to show that M. tuberculosis peptide–MHC class II complexes are formed in phagosomes.
Majlessi, L. et al. Inhibition of phagosome maturation by mycobacteria does not interfere with presentation of mycobacterial antigens by MHC molecules. J. Immunol. 179, 1825–1833 (2007).
Ramachandra, L. et al. Phagosomal processing of Mycobacterium tuberculosis antigen 85B is modulated independently of mycobacterial viability and phagosome maturation. Infect. Immun. 73, 1097–1105 (2005).
Gonzalez-Juarrero, M., Shim, T. S., Kipnis, A., Junqueira-Kipnis, A. P. & Orme, I. M. Dynamics of macrophage cell populations during murine pulmonary tuberculosis. J. Immunol. 171, 3128–3135 (2003).
Humphreys, I. R. et al. A role for dendritic cells in the dissemination of mycobacterial infection. Microbes Infect. 8, 1339–1346 (2006).
Pecora, N. D. et al. Mycobacterium bovis BCG decreases MHC-II expression in vivo on murine lung macrophages and dendritic cells during aerosol infection. Cell. Immunol. 254, 94–104 (2008).
Reljic, R. et al. Time course of mycobacterial infection of dendritic cells in the lungs of intranasally infected mice. Tuberculosis 85, 81–88 (2005).
Banchereau, J. & Steinman, R. M. Dendritic cells and the control of immunity. Nature 392, 245–252 (1998).
Winzler, C. et al. Maturation stages of mouse dendritic cells in growth factor-dependent long-term cultures. J. Exp. Med. 185, 317–328 (1997).
Cella, M., Engering, A., Pinet, V., Pieters, J. & Lanzavecchia, A. Inflammatory stimuli induce accumulation of MHC class II complexes on dendritic cells. Nature 388, 782–787 (1997). This article demonstrates that the mechanism for increased MHC class II molecule expression on DC maturation involves enhanced stability of MHC class II molecules, whereas protein synthesis is only transiently increased. This reveals that the primary mechanism for maintaining enhanced MHC class II molecule expression on DCs is post-translational rather than transcriptional.
De Smedt, T. et al. Regulation of dendritic cell numbers and maturation by lipopolysaccharide in vivo. J. Exp. Med. 184, 1413–1424 (1996).
Askew, D., Chu, R. S., Krieg, A. M. & Harding, C. V. CpG DNA induces maturation of dendritic cells with distinct effects on nascent and recycling MHC-II antigen processing mechanisms. J. Immunol. 165, 6889–6895 (2000).
Shin, J. S. et al. Surface expression of MHC class II in dendritic cells is controlled by regulated ubiquitination. Nature 444, 115–118 (2006).
van Niel, G. et al. Dendritic cells regulate exposure of MHC class II at their plasma membrane by oligoubiquitination. Immunity 25, 885–894 (2006). References 124 and 125 reveal that regulation of MHC class II molecule stability and expression in DCs involves regulation of MHC class II molecule ubiquitylation, providing a mechanism for the regulation of MHC class II molecule expression in the context of DC maturation.
De Gassart, A. et al. MHC class II stabilization at the surface of human dendritic cells is the result of maturation-dependent MARCH I down-regulation. Proc. Natl Acad. Sci. USA 105, 3491–3496 (2008). This work shows that a ubiquitin E3 ligase, MARCHI, is down-regulated during dendritic cell maturation, clarifying the mechanism for ubiquitin-mediated control of MHC class II stability and expression in dendritic cells.
Landmann, S. et al. Maturation of dendritic cells is accompanied by rapid transcriptional silencing of class II transactivator (CIITA) expression. J. Exp. Med. 194, 379–391 (2001).
Hava, D. L. et al. Evasion of peptide, but not lipid antigen presentation, through pathogen-induced dendritic cell maturation. Proc. Natl Acad. Sci. USA 105, 11281–11286 (2008).
Murray, R. A., Siddiqui, M. R., Mendillo, M., Krahenbuhl, J. & Kaplan, G. Mycobacterium leprae inhibits dendritic cell activation and maturation. J. Immunol. 178, 338–344 (2007).
Uehori, J. et al. Dendritic cell maturation induced by muramyl dipeptide (MDP) derivatives: monoacylated MDP confers TLR2/TLR4 activation. J. Immunol. 174, 7096–7103 (2005).
Tsuji, S. et al. Maturation of human dendritic cells by cell wall skeleton of Mycobacterium bovis bacillus Calmette-Guéerin: involvement of Toll-like receptors. Infect. Immun. 68, 6883–6890 (2000).
Inaba, K., Inaba, M., Naito, M. & Steinman, R. M. Dendritic cell progenitors phagocytose particulates, including bacillus Calmette-Guerin organisms, and sensitize mice to mycobacterial antigens in vivo. J. Exp. Med. 178, 479–488 (1993).
Tailleux, L. et al. DC-SIGN is the major Mycobacterium tuberculosis receptor on human dendritic cells. J. Exp. Med. 197, 121–127 (2003).
Jiao, X. et al. Dendritic cells are host cells for mycobacteria in vivo that trigger innate and acquired immunity. J. Immunol. 168, 1294–1301 (2002).
Pedroza-Gonzalez, A. et al. In situ analysis of lung antigen-presenting cells during murine pulmonary infection with virulent Mycobacterium tuberculosis. Int. J. Exp. Pathol. 85, 135–145 (2004).
Ordway, D., Henao-Tamayo, M., Orme, I. M. & Gonzalez-Juarrero, M. Foamy macrophages within lung granulomas of mice infected with Mycobacterium tuberculosis express molecules characteristic of dendritic cells and antiapoptotic markers of the TNF receptor-associated factor family. J. Immunol. 175, 3873–3881 (2005).
Hamerman, J. A. & Aderem, A. Functional transitions in macrophages during in vivo infection with Mycobacterium bovis bacillus Calmette-Guerin. J. Immunol. 167, 2227–2233 (2001).
Krutzik, S. R. & Modlin, R. L. The role of Toll-like receptors in combating mycobacteria. Semin. Immunol. 16, 35–41 (2004).
Coulombe, F. et al. Increased NOD2-mediated recognition of N-glycolyl muramyl dipeptide. J. Exp. Med. 206, 1709–1716 (2009).
Pandey, A. K. et al. NOD2, RIP2 and IRF5 play a critical role in the type I interferon response to Mycobacterium tuberculosis. PLoS Pathog. 5, e1000500 (2009).
Wearsh, P. & Cresswell, P. Antigen processing and presentation. Poster. Nature Rev. Immunol. 9 (2009).
Drage, M. G. et al. Differences in expression of TLR2 and its co-receptors determine responses of antigen presenting cells to lipoproteins of Mycobacterium tuberculosis. Cell. Immunol. 258, 29–37 (2009).
Acknowledgements
Research in the authors' laboratories is supported by US National Institutes of Health grants AI035726, AI034343 and AI069085 to C.V.H. and grants HL055967 and AI027243 to W.H.B.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Related links
Related links
DATABASES
Entrez Genome Project
Mycobacterium bovis bacille Calmette—Guérin
FURTHER INFORMATION
Glossary
- CD4+ T cell
-
A T cell that expresses the CD4 receptor. These cells recognize antigens that are presented on the surface of host cells by MHC class II molecules.
- CD8+ T cell
-
A T cell that expresses the CD8 receptor. These cells recognize antigens that are presented on the surface of host cells by MHC class I molecules, leading to host cell destruction, and are therefore also known as cytotoxic T cells.
- γδ T cell
-
A T cell that expresses the γδ T cell receptor. Although the exact function of γδ T cells is unknown, it has been suggested that mucosal γδ T cells are involved in innate immune responses
- Granuloma
-
An organized structure that comprises lymphocytes, macrophages, neutrophils and, sometimes, fibroblasts and that often has a necrotic centre, which arises in response to continued antigenic stimulation in the presence of macrophages (as occurs, for example, in M. tuberculosis infection).
- Toll-like receptor
-
A membrane-spanning protein that recognizes conserved ligands on pathogens, such as flagellin, lipopolysaccharide or DNA, and that is therefore a key recognition molecule in the host innate immune response.
- Tuberculin skin test
-
A method of diagnosing M. tuberculosis infection by injecting TB antigens intradermally. A delayed-type hypersensitivity response, dependent on the presence of sensitized T cells, is seen in those infected with M. tuberculosis. This does not distinguish latent infection from active TB.
- Pathogen-associated molecular pattern
-
A small molecular motif that is conserved across microbial species and engages innate immune receptors, in particular TLRs. Examples include lipopolysaccharide, peptidoglycan and flagellin.
- Natural killer cell
-
A lymphocyte that does not express the T cell receptor or B cell receptor and that confers innate immunity.
- T helper 2 cell
-
A type of activated T helper cell that participates in phagocytosis-independent responses and downregulates pro-inflammatory responses that are induced by T helper 1 cells. T helper 2 cells secrete IL-4, IL-5 and IL-6.
- Regulatory T cell
-
A CD4+ T cell that naturally expresses high levels of CD25 (the IL-2 receptor subunit-α) and the transcription factor forkhead box P3 (FOXP3) and that has suppressive regulatory activity towards effector T cells and other immune cells.
- Pyogenic bacterium
-
A pus-forming bacterium that is associated with exudative inflammation and neutrophil recruitment.
Rights and permissions
About this article
Cite this article
Harding, C., Boom, W. Regulation of antigen presentation by Mycobacterium tuberculosis: a role for Toll-like receptors. Nat Rev Microbiol 8, 296–307 (2010). https://doi.org/10.1038/nrmicro2321
Issue Date:
DOI: https://doi.org/10.1038/nrmicro2321
This article is cited by
-
C-terminal region of Rv1039c (PPE15) protein of Mycobacterium tuberculosis targets host mitochondria to induce macrophage apoptosis
Apoptosis (2024)
-
Association of TLR4 and TLR9 gene polymorphisms with cervical HR-HPV infection status in Chinese Han population
BMC Infectious Diseases (2023)
-
Activation dynamics of antigen presenting cells in vivo against Mycobacterium bovis BCG in different immunized route
BMC Immunology (2023)
-
Role of C-terminal domain of Mycobacterium tuberculosis PE6 (Rv0335c) protein in host mitochondrial stress and macrophage apoptosis
Apoptosis (2023)
-
Decoding the spatial chromatin organization and dynamic epigenetic landscapes of macrophage cells during differentiation and immune activation
Nature Communications (2022)