Abstract
Background
There is substantial evidence that heart rate (HR) is a powerful predictor of mortality in both normal individuals and in patients with cardiovascular disease. The use of b-adrenoceptor antagonists (β-blockers) has confirmed the importance of lowering elevated HR in a patient’s prognosis. However, these agents can have undesirable adverse effects (AEs) and due to the risk of bronchoconstriction are contraindicated in patients with obstructive airway disease. A selective bradycardic agent, without such undesirable effects, could be of therapeutic interest. Ivabradine, a new I f inhibitor that acts specifically on the sino-atrial node, is a pure HR-lowering agent.
Objective
The objective of this study was to assess HR-lowering efficacy and respiratory safety of ivabradine in patients with asthma and chronic obstructive pulmonary disease (COPD).
Methods
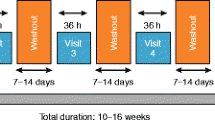
This was a randomized, single-center, double-blind, placebo-controlled, crossover trial. Enrolment began in May 2009, and the last patient completed the study in January 2011. The study was conducted in an ambulatory setting. A total of 40 patients completed the study (20 asthmatic patients and 20 COPD patients). Inclusion criteria were: documented diagnosis of asthma or COPD according to international guidelines, age 18–75 years, and mean HR on Holter ECG recording of ≥60 beats/min. Exclusion criteria included disease exacerbation in a previous month or inability to understand instructions on the study procedures. All patients received ivabradine 7.5 mg twice daily for 5 days and placebo twice daily for 5 days in a crossover manner, in one of the two arms of the study, with at least 2 days of washout between treatments. The main outcome measures included the difference in HR between ivabradine and placebo treatment and change in HR in comparison with baseline. Other evaluated outcomes were differences in the peak expiratory flow rate (PEFR), the daily symptom score, rescue medication consumption, and AEs. Results: Ivabradine produced significantly lower mean HR than placebo in both groups of patients: asthma 67.4 ± 8.38 versus 82.85±11.19 beats/min (p<0.001) and COPD 69.75 ± 8.9 versus 81.05 ± 9.75 beats/min (p<0.001). Similar results were observed for the minimal HR as well as for the maximal noted HR. In comparision with baseline, ivabradine significantly reduced HR in both groups of studied patients (all p < 0.05), whereas placebo did not have such an effect. No significant difference, in either the asthma or the COPD group, was found between ivabradine and placebo in morning and evening peak expiratory flow rate, peak expiratory flow diurnal variability, daily symptom scores, and rescue medication usage (all p > 0.05). Both treatments were well tolerated. The incidence of AEs was low and generally similar in both periods of treatment, except for visual symptoms during treatment with ivabradine, which was reported by 5% of the patients.
Conclusion
Our study demonstrated that selective HR reduction with ivabradine is effective in patients with asthma and COPD, with no alteration in respiratory function or symptoms over the duration of the study. Ivabradine offers an interesting alternative, as an HR-lowering agent, in patients with respiratory disease and contraindications to β-blockers.
Clinical Trial Registration
Registered at www.clinicaltrials.gov (NCT01365286).









Similar content being viewed by others
References
Kannel WB, Kannel C, Paffenbarger RS, et al. Heart rate and cardiovascular mortality: the Framingham Study. Am Heart 1987; 113: 1489–94
Palatini P, Casiglia E, Pauletto P, et al. Relationship of tachycardia with high blood pressure and metabolic abnormalities: a study with a mixture analysis in three populations. Hypertens 1997; 30: 1267–73
Hjalmarson A, Gilpin EA, Kjekshus G, et al. Influence of heart rate on mortality after acute myocardial infarction. Am J Cardiol 1990; 1: 547–53
Kjekshus J, Gullestad L. Heart rate as a therapeutic target in heart failure. Eur Heart J Supplements 1999; 1(Suppl. H): H64–9
Gillum RF, Makuc DM, Feldman JJ. Pulse rate, coronary heart disease, and death: the NHANES I Epidemiologic Follow-up Study. Am Heart J 1991; 121 no. 1: 172–7
Fox K, Borer JS, Camm AJ, et al. Resting heart rate in cardiovascular disease. J Am Coll Cardiol 2007 Aug 28; 50(9): 823–30
Freemantle N, Cleland J, Young P, et al. Beta blockade after myocardial infarction: systematic review and meta regression analysis. BMJ 1999; 318: 1730–7
Frishman WH, Furberg CD, Friedewald WT. The use of beta-adrenergic blocking drugs in patients with myocardial infarction. Curr Probl Cardiol 1984; 9: 1–50
Gheorghiade M, Goldstein S. Beta-blockers in the post-myocardial infarction patient. Circulation 2002; 4: 394–8
Farrell MH, Foody JM, Krumholz HM. Beta-blockers in heart failure: clinical applications. JAMA 2002; 7: 890–7
Fihn SD, Williams SV, Daley J, et al. Guidelines for the management of patients with chronic stable angina: treatment. Ann Intern Med 2001; 135(8 Pt 1): 616–32
Benson MK, Berrill WT, Cruickshank JM, et al. A comparison of four betaadrenoceptor antagonists in patients with asthma. Br J Clin Pharmacol 1978; 5: 415–9
Schwartz S, Davies S, Juers JA. Life-threatening cold and exercise induced asthma potentiated by administration of propranolol. Chest 1980; 78: 100–1
Wiliams IP, Millard FJC. Severe asthma after inadvertent ingestion of oxprenolol. Thorax 1980; 35: 160
Chang CL, Mills GD, McLachlan JD, et al. Cardio-selective and non-selective beta-blockers in chronic obstructive pulmonary disease: effects on bronchodilator response and exercise. Intern Med J. 2010 Mar; 40(3): 193–200
DiFrancesco D, Camm JA. Heart rate lowering by specific and selective I(f) current inhibition with ivabradine: a new therapeutic perspective in cardiovascular disease. Drugs 2004; 64: 1757–65
Tendera M. If inhibition: from pure heart rate reduction to treatment of stable angina. Eur Heart J Suppl. 2005; 7 Suppl. H: H3–6
Babu KS, Gadzik F, Holgate ST. Absence of respiratory effects with ivabradine in patients with asthma. Br J Clin Pharmacol 2008 Jul; 66(1): 96–101
Ageev FT, Makarova GV, Patrusheva IF, et al. The efficacy and safety of the combination of β-blocker bisoprolol and I(f) inhibitor ivabradine in patients with stable angina and chronic obstructive pulmonary disease. Kardiologiia 2010; 50(10): 22–6
Beasley R. The global burden of asthma report, Global Initiative for Asthma (GINA) 2004 [online]. Available from URL: http://www.ginasthma.org [Accessed 2011 Sep 7]
Global Strategy for the Diagnosis, Management and Prevention of COPD, Global Initiative for Chronic Obstructive Lung Disease (GOLD) 2010 [online]. Available from URL: http://www.goldcopd.org [Accessed 2011 Sep 7]
Kjekshus JK. Importance of heart rate in determining beta-blocker efficacy in acute and long-term myocardial infarction intervention trials. Am J Cardiol 1986; 57: 43F-9F
Lechat P. Beta-blocker treatment in heart failure: role of heart rate reduction. Basic Res Cardiol 1998; 93 Suppl. 1: 148–55
Fox K, Garcia MA, Ardissino D, et al. Guidelines on the management of stable angina pectoris: executive summary. The Task Force on the Management of Stable Angina Pectoris of the European Society of Cardiology. Eur Heart J 2006; 27: 1341–81
Ford ES, Giles WH, Croft JB. Prevalence of nonfatal coronary heart disease among American adults. Am Heart J 2000; 139: 371–7
Lipworth BJ, Irvine NA, McDevitt DG. The effects of chronic dosing on the beta 1 and beta 2-adrenoceptor antagonism of betaxolol and atenolol. Eur J Clin Pharmacol. 1991; 40(5): 467–71
Lammers JW, Folgering HT, van Herwaarden CL. Respiratory tolerance of bisoprolol and metoprolol in asthmatic patients. J Cardiovasc Pharmacol 1986; 8 Suppl. 11: S69–73
Dickstein K, Cohen-Solal A, Filippatos G, et al. ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure 2008: the Task Force for the Diagnosis and Treatment of Acute and Chronic Heart Failure 2008 of the European Society of Cardiology. Developed in collaboration with the Heart Failure Association of the ESC (HFA) and endorsed by the European Society of Intensive Care Medicine (ESICM). Eur Heart J 2008; 29: 2388–442
Hunt SA, Abraham WT, Chin MH, et al. ACC/AHA 2005 Guideline Update for the Diagnosis and Management of Chronic Heart Failure in the Adult: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to Update the 2001 Guidelines for the Evaluation and Management of Heart Failure): developed in collaboration with the American College of Chest Physicians and the International Society for Heart and Lung Transplantation: endorsed by the Heart Rhythm Society. Circulation 2005; 112: e154–235
Salpeter S, Ormiston T, Salpeter E. Cardioselective beta-blockers for reversible airway disease. Cochrane Database Syst Rev 2002; (4): CD002992
Salpeter SR, Ormiston TM, Salpeter EE, et al. Cardioselective beta-blockers for chronic obstructive pulmonary disease: a meta-analysis. Respir Med 2003 Oct; 97(10): 1094–101
Dransfield MT, Rowe SM, Johnson JE, et al. Use of beta blockers and the risk of death in hospitalised patients with acute exacerbations of COPD. Thorax 2008 Apr; 63(4): 301–5
van Gestel YR, Hoeks SE, Sin DD, et al. Impact of cardioselective betablockers on mortality in patients with chronic obstructive pulmonary disease and atherosclerosis. Am J Respir Crit Care Med 2008 Oct 1; 178(7): 695–700
Short PM, Lipworth SI, Elder DH, et al. Effect of beta blockers in treatment of chronic obstructive pulmonary disease: a retrospective cohort study. BMJ 2011 May 10; 342: d2549.doi: 10.1136/bmj.d2549
Baker JG. The selectivity of beta-adrenoceptor antagonists at the human beta1, beta2 and beta3 adrenoceptors. Br J Pharmacol 2005 Feb; 144(3): 317–22
van der Woude HJ, Zaagsma J, Postma DS, et al. Detrimental effects of betablockers in COPD: a concern for nonselective b-blockers. Chest 2005; 127: 818–24
Lee DK, Borade PS, Currie GP. Safety of beta-blockers in COPD: the jury must still be out. QJM 2005 Sep; 98(9): 706–7
Albouaini K, Andron M, Alahmar A, et al. Beta-blockers use in patients with chronic obstructive pulmonary disease and concomitant cardiovascular conditions. Int J Chron Obstruct Pulmon Dis 2007; 2: 535–40
Egred M, Shaw S, Mohammad B, et al. Under-use of beta-blockers in patients with ischaemic heart disease and concomitant chronic obstructive pulmonary disease. Q J Med 2005; 98: 493–7
Brooks TW, Creekmore FM, Young DC, et al. Rates of hospitalizations and emergency department visits in patients with asthma and chronic obstructive pulmonary disease taking beta-blockers. Pharmacotherapy 2007 May; 27(5): 684–90
Masoli M, Fabian D, Holt S, et al. The global burden of asthma: executive summary of the GINA Dissemination Committee report. Allergy 2004; 59(5): 469–78
Roche N, Huchon G. Epidemiology of chronic obstructive pulmonary disease. Rev Prat 2004 Sep 15; 54(13): 1408–13
Antonelli Inclazi R, Fuso L, De Rosa M, et al. Co-morbidity contributes to predict mortality of patients with obstructive pulmonary disease. Eur Respir J 1997; 10: 2794–800
Cazzola M, Calzetta L, Bettoncelli G, et al. Asthma and comorbid medical illness. Eur Respir J 2011 Jul; 38(1): 42–9
Salpeter SR, Ormiston TM, Salpeter EE. Cardiovascular effects of betaagonists in patients with asthma and COPD: a meta-analysis. Chest 2004 Jun; 125(6): 2309–21
Sears MR. Adverse effects of beta-agonists. J Allergy Clin Immunol 2002; 110: S322–8
Fox K, Ford I, Steg PG, et al. Ivabradine for patients with stable coronary artery disease and left ventricular systolic dysfunction (BEAUTIFUL): a randomised, double-blind, placebo-controlled trial. Lancet 2008 Sep 6; 372(9641): 807–16
Borer JS, Fox K, Jaillon P, et al. Antianginal and antiischemic effects of ivabradine, an I(f) inhibitor, in stable angina: a randomized, double-blind, multicentered, placebo-controlled trial. Circulation 2003; 107: 817–23
Tardif JC, Ford I, Tendera M, et al. Efficacy of ivabradine, a new selective I(f) inhibitor, compared with atenolol in patients with chronic stable angina. Eur Heart J 2005; 26: 2529–36
Swedberg K, Komajda M, Böhm M, et al. Ivabradine and outcomes in chronic heart failure (SHIFT): a randomised placebo-controlled study. Lancet 2010 Sep 1; 376(9744): 875–85
Murata GH, Lium DJ, Busby HK, et al. Precision and accuracy of self-measured peak expiratory flow rates in chronic obstructive pulmonary disease. South Med J 1998 Oct; 91(10): 919–24
Murata GH, Kapsner CO, Lium DJ, et al. Patient compliance with peak flow monitoring in chronic obstructive pulmonary disease. Am J Med Sci 1998 May; 315(5): 296–301
Szafranski W, Cukier A, Ramirez A, et al. Efficacy and safety of budesonide/formoterol in the management of chronic obstructive pulmonary disease. Eur Respir J 2003 Jan; 21(1): 74–81
Llewellin P, Sawyer G, Lewis S, et al. The relationship between FEV1 and PEF in the assessment of the severity of airways obstruction. Respirology 2002 Dec; 7(4): 333–7
Kaneko A, Tachibana M. A voltage-clamp analysis of membrane currents in solitary bipolar cells dissociated from Carassius aureus. J Physiol 1985; 358: 131–52
Demontis GC, Longoni B, Barcaro U, et al. Properties and functional roles of hyperpolarization-gated currents in guinea-pig retinal rods. J Physiol 1999; 515: 813–28
Satoh TO, Yamada M. A bradycardiac agent ZD7288 blocks the hyperpolarization-activated current (I(h)) in retinal rod photoreceptors. Neuropharmacology 2000; 39: 1284–91
Acknowledgments
The study was funded by the Medical University of Lodz, Poland. The authors’ work was independent of the funder. None of the authors have any conflicts of interest to declare.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Majewski, S., Slomka, S., Zielinska-Wyderkiewicz, E. et al. Heart Rate-Lowering Efficacy and Respiratory Safety of Ivabradine in Patients with Obstructive Airway Disease. Am J Cardiovasc Drugs 12, 179–188 (2012). https://doi.org/10.2165/11597400-000000000-00000
Published:
Issue Date:
DOI: https://doi.org/10.2165/11597400-000000000-00000